THIS TEXT IS CONTINUOUSLY EDITED, CORRECTED, EXPANDED, AND UPDATED
In this post, we discuss the diversity of obligate troglobitic, troglobiont, cave-dewlling, obligatory subterranean, and stygobitic animals in Brazil, collectively referred to here by the term troglobic (see discussion below). A comprehensive literature review was conducted, with a strong focus on the global context, and as of May 14, 2026, 893 troglobic species have been recorded in Brazil and 9,499 in World. Brazilian diversity span over 8 of the 11 Metazoa elegible phyla for troglobic: Porifera (2), Platyhelminthes (18), Nemertea (3), Annelida (9), Mollusca (31), Onychophora (1), Arthropoda (790) and Craniata (39). Each species is presented in own card, featuring distribution map and images, when available. We also conducted cross-sectional analyses, examining the literature on the distribution of these species in various regions of the world, as well as their occurrence by state, municipality, and taxonomic group in Brazil.
This study, in the Brazilian totals and in some global totals, chose to include undescribed species. In the national case, more than 500 species cited in 30 articles as troglobitic and/or troglomorphic were compiled, many of them in a preliminary manner.
It is important to emphasize that this decision is highly unstable, as undescribed species may later prove to be synonyms of already described taxa, may not in fact be troglobitic after detailed analysis, may belong to a different group than originally cited, or may represent the same species cited in different articles. Additionally, there is the inherent conceptual difficulty in defining troglobiontism.
In this text, species are referred to by the names used in their original publications, and care will be taken to update this list should any of them be formally described in the future. Their global status will be corrected accordingly, and any species shown not to be truly troglobitic will be excluded. Details of these more than 500 undescribed species are provided in Section 6 of this work.
1 CONVENTIONS OF THIS TEXT
For nomenclature, we use the notation ( z : y / ) x, which refers to x species in y genera of z families.
2 DEFINITIONS OF CAVE-OBLIGATORY ANIMALS
2A CLASSIFICATION OF SUBTERRANEAN SPECIES
For the purposes of this text, based on Trajano & Bichuette (Biologia Subterrânea/Introdução, 2006), we will use the following terminology for Metazoarian in caves: (1) troglobionts — obligatory subterranean, exclusively subterranean source populations; sink populations may be found in surface habitats; if obligate subterranean aquatic, we will call stygobionts; (2) troglophiles are subterranean source populations that maintain genetic connection with the epigean ones through individuals that disperse between these habitats; (3) trogloxenes are individuals regularly found in subterranean habitats but which must return periodically to the surface to complete their life cycle; they would be instances of source populations in epigean habitats, but using subterranean resources (the obligatory trogloxenes, all individuals are dependent on both subterranean and surface resources).
2B NOMENCLATURAL ADJUSTMENT
The terms listed above for classifying subterranean species are confusing, and in many contexts, they end up being inefficient in communication between different sources. Aiming to standardize the language used in this post, we have adopted the following terms to represent the different groups of obligatorily subterranean animals:
troglobic → obligatory subterranean, with exclusively subterranean source populations; sink populations may be found in surface habitats. Used here generically for terrestrial and freshwater species.
troglobiont → terrestrial troglobic, used in the text if necessary.
stygobitic → freshwater or amphibious troglobic, used in the text if necessary.
halotroglobic → marine cave-obligatory animals, no-troglobic in this work.
The Brazilian list of troglobic includes subterranean species from caves, both terrestrial and freshwater, as well as phreatic fish species. It excludes hypotelminorheic species (there is at least one in Brazil, in Rio Grande do Sul state) and marine species, which are entirely absent from Brazilian marine caves, as the country unfortunately lacks anchialine caves.
2C CRITERIA FOR TROGLOBIC STATUS
When discussing the concept of troglobic, four key questions arise for analysis. The first concerns whether this concept also applies to fungi and other organisms, such as protists, which are traditionally classified separately. The second question is defining which habitats qualify a potential troglobic as a true troglobic: anchialine caves, littoral interstitial habitats, deep phreatic and hyporheic of rivers, MSS (milieu souterrain superficiel, underground network of empty air-filled voids and cracks developing within multiple layers of rock fragments, generally covered by topsoil, Naturwissenschaften, 2016), epikarts and hypotelminorheic habitats can be? The third issue involves defining troglobic within well-established biological groups, exploring apomorphies (derived characteristics) and the phenomenon of troglomorphism, which refers to adaptations specific to subterranean environments. Finally, the fourth point questions to what extent the concept of troglobic can be applied to marine organisms, considering the unique features of aquatic environments and their caves. Culver, Deharveng, Bedos & Pipan (Diversity, 2021) makes interesting observations about some of these points.
Troglobic are usually recognized by the presence of troglomorphisms - autapomorphies related to the subterranan selective regime. The commonest and most conspicuous troglomorphisms are reduction up to complete loss of visual structures and dark tegumentar pigmentation; other frequent troglomorphisms include elongation of barbels in fish and of appendices in arthropods, and reduction of wings in insects.
The classification of a species as troglobic is a more subjective than objective assessment. Over the decades, different criteria have been proposed and adopted, varying according to the taxonomic group and the author. The same species may receive contradictory status depending on the source or checklist consulted. In this blog, we follow the classification adopted by the authors — almost always those who originally described the species — intervening only in a few cases where the authors themselves were unable to determine the organism’s true ecological status. Simões, Souza-Silva, Venâncio & Ferreira (9th International Congress of Speleology, 2025) features a noteworthy discussion of this issue, outlining several criteria used to classify a species as troglobitic and how these criteria vary across the main taxonomic groups.
INCLUSION
I. Low probability of survival in the epigean habitat, even if there are no apparent troglomorphisms.
II. Obvious troglomorphisms and the distribution is not wide and/or unconnected.
EXCLUSION
I. Existence of populations in the epigean environment.
II. No apparent troglomorphisms and a high probability of survival in the epigean environment.
NEED FOR ADDITIONAL STUDIES
I. Group with difficulties in determining troglomorphisms.
II. Obvious troglomorphisms, but distribution is wide and/or unconnected.
2D ARE PARASITES OF TROGLOBIC HOSTS AUTOMATICALLY TROGLOBIC?
Could parasitic species found exclusively on troglobic hosts also be considered troglobic? Would their dependence represent a unique adaptation to subterranean environments? There is little discussion on this topic, and few studies address it. The list of troglobic from the United States and Canada[33] includes species from at least three phyla, parasitic on troglobic species, as troglobic. Here, we follow this approach, although a more in-depth discussion on the subject is still needed.
2E TROGLOPHILIC TAXA
An excellent and important study on the troglophilic fauna of Brazil, which is neglected in conservation measures, is Duarte, Gallão & Bichuette (Biota Neotropica, 2025), citing 223 described troglophilic species, distributed across 51 orders and five phyla.
2F GROUPS EXCLUDED FROM THE CONCEPT OF TROGLOBIC
The scope of this post is limited exclusively to obligatory subterranean freshwater and terrestrial species. A large number of references extend the term stygobiont to species inhabiting marine environments, especially anchialine systems. These are not considered here.
TAXONOMY OF EXCLUDED TAXA
Accordingly, we excluded as much data as possible on Stygiomysida (Mexico, Bermuda, among others), Thecostraca/Tantulocarida (Stygotantulus stocki Boxshall & Huys, 1989, which was reported from anchialine environments of the lava tubes in the Canary Island, parasitizing two families of Copepoda), Cumacea (subterranean species are collected from Bermudas, Bahamas and Jamaica), Tanaidacea (marine benthic habitats around the world, inhabiting anchialine and marine caves as halotroglobic in different regions of the world such as Bermuda and several islands of the South Pacific), Remipedia (25, Mexico, Belize, Caribbean, Canary Island, Australia), Stenopodida (1, Bahamas), Gebiida (1, Bahamas), Mictacea (a monotypic order with a halotroglobic, described for marine caves of Bermuda), Bochusacea (deep sea benthos, but includes two halotroglobic species of anchialine caves of the Bahamas) and Leptostraca (1, Providenciales caves in the Turks y Caicos), whose cave-restricted forms are entirely marine, as well as the marine forms of Anomura (1, Canary Is.), Amphipoda (Mexico, Bermuda, among others), Isopoda (Mexico, Bermuda, among others), Copepoda (Mexico, Bermuda, among others), Ostracoda (Mexico, Bermuda, among others), Mysida (Mexico, Bermuda, among others), Thermosbaenacea (includes freshwater troglobic and some halotroglobic worldwide), Caridea (Mexico, Bermuda, Honduras, Bahamas, among others, SEE) and Brachyura (no data).
All troglobic species in Branchipoda (Europe), Anaspidacea (Australia, New Zealand, Argentina and Chile), Astacidea (40, Papua New Guinea, USA, Mexico and Cuba), Spelaeogriphacea (W Brazil, South Africa and W Australia) and Bathynellacea (widely worldwide in subterranean habitats) are fully freshwaters.
Six infraorders of Decapoda include subterranean forms, with two of them consisting exclusively of marine species: Stenopodidae, represented by a species of the Macromaxillocarididae family from a anchialine cave in Bahamas; and Gebiidea, with a singles anchialine species of the genus Naushonia (Laomediidae). also for the Bahamas. One Anomura is a halotroglobic, in Canary Is. (Munidopsis polymorpha Koelbel, 1892).
All (8:12/)28 spp. of Remipedia are halotroglobic anchialine (SEE), in Bahamas (7/13), Turks y Caicos (4/4), Mexico (1/3), Canary Is. (1/2), Australia (1/2), Republica Dominicana (1/2) and Cuba (1/2). Kaloketos, Lasionectes and Micropacteris occur only in Turks y Caicos. Angirasu, Cryptocorynetes, Godzilliognomus and Pleomothra occur only in Bahamas Godzillius in these both areas Other genera are Speleonectes (5, 4 in Bahamas, one in Cuba), Morlockia (4, Bahamas and Rep. Dominicana one each, 2 in Canary Is., Spain), Kumonga (1, NW Australia), and Xibalbanus (4, SE Mexico and Belize).
ANCHIALINE SYSTEMS
An anchialine ecosystem is defined as 'a tidally-influenced subterranean estuary located within crevicular and cavernous karst and volcanic terrains that extends inland to the limit of seawater penetration' (Calderón-Gutiérrez et al., Regional Studies in Marine Science, 2017). For geral data from species in anchialine caves, see Perez-Moreno et al. (International Journal of Speleology, 2016).
MEXICAN ANCHIALINE
Up to now, a total of 67 anchialine species have been recorded in anchialine caves from Mexico, all in Yucatan Peninsula, including organisms belonging to six phyla: Porifera, Annelida, Crustacea, Mollusca, Echinodermata, and Chordata. All 10 species of anchialine Porifera are all from Cozumel Island occurring so far, in only two caves: La Quebrada and Aerolito. One Annelida of the genus Speleonerilla (Nerillidae) from two cenotes in the Yucatan Peninsula is reported - however, they did not describe a new species due to the limited number of individuals available. Arthropoda represent the most diverse group in the anchialine caves of Mexico, with (23:36/)51 spp. that belong to 4 classes and 9 orders, with one endemic family (Anchialocarididae) and eight endemic genera (Mexicophria, Mayaweckelia, Tuluweckelia, Creaseriella, Yucatalana, Yagerocaris, Anchialocaris, Creasaria), plus 44 endemic species, in Remipedia (3), Melascostraca - Decapoda (16), Amphipoda (6), Mysida (3), Isopoda (7), Thermosbaenacea (1), Copepoda (12), and Ostracoda (3). Up to now, one species of anchialine Mollusca has been described from Mexico, the gastropod Teinostoma brankovitsi (Tornidae). Echinodermata anchialine fauna of Mexico includes Copidaster cavernicola, has been recorded so far from cenote Aerolito in Cozumel, and six undescribed species of asteroids and ophiuroids, all from cenote Aerolito that await formal description and always in sections next to connections to the sea due to marine nature from Echinodermata (Scientific Reports). Two species of Chordata (both Actinopteri), the blind cusk eel Typhlias pearsei (Dinematichthydae), and the blind eel Ophisternon infernale (Synbranchidae) occur in the anchialine caves of the Yucatan Peninsula, always in the freshwater layer (Alvarez et al., Mexican Fauna in the Anthropocene, 2023).
From the troglobic accepted here in the Mexican troglofauna, we exclude anchialine species: Stygiomysis (2, Stygiomysida), Danielopolina mexicana Kornicker & Iliffe (Ostracoda), Agostocaris bozanici Kensley (Decapoda, Caridea), Triacanthoneus akumalensis Álvarez, Iliffe, González, & Villalobos (Decapoda, Caridea), Yagerocaris cozumel Kensley (Decapoda, Caridea), Barbouria yanezi Mejía, Zarza, & López (Decapoda, Caridea), Calliasmata nohochi Escobar-Briones, Camacho & Alcocer (Decapoda, Caridea), Procaris mexicana von Sternberg & Schotte (Decapoda, Procaridea) by [32], Xibalbanus (3, Remipedia), and Tulumella unidens Bowman & Iliffe, 1988 (Thermoasbaenacea) in Mexico, totaling 13 species (World Remipedia Database | Proceedings of the Biological Society of Washington, 1998).
Five Mexican anchialine caves have more than 10 species, El Aerolito system has 100 (Calderón-Gutiérrez et al., Regional Studies in Marine Science, 2017). For Porifera in anchialine caves in Mexico, see (Goméz et al., Zootaxa, 2020). Alvarez et al. (Mexican Fauna in the Anthropocene, 2023) cites 67 anchialine species have been described in SE Mexico, 58 of which are endemic to the region. For aditional data for anchialine fauna distributed within a cave (Ox Bel Ha, SE Mexico), see Benítez et al. (Subterranean Biology, 2019).
BERMUDA ANCHIALINE
The fauna inhabiting caves in the Walsingham Tract in Bermuda consists of 78 described species of cave-dwelling invertebrates, including 63 stygobionts and 15 stygophiles. Thus, it represents one of the world’s top hotspots of subterranean biodiversity. Of the anchialine fauna,
65 of the 78 species are endemic to Bermuda, while 66 of the 78 are crustaceans (Iliffe & Gutiérrez, Diversity, 2021). The text cites 63 stygobitics, but its table lists only 62, and excluding the questionable records of two Ciliophora, we conclude with 60 stygobic for the site: Annelida (3), Gastropoda (2), Acari (5), Copepoda (16), Ostracoda (13), Caridea (5), Isopoda (4), Amphipoda (5), Ingolfiellida (1), Tanaidacea (1), Mictacea (1), Cumacea (2), and Mysida (2).
VENEZUELA
In Venezuela, so far, only two endemic anchialine species, Cyathura univam Botosaneanu 1983 (Isopoda: Anthuridea) and Metaniphargus venezuelanus Stock & Botosaneanu 1983 (Amphipoda: Hadziidae), have been reported, and they were described from specimens collected in a cave not yet officially recorded by professional speleologists in Falcon state, specifically in the Mallorquines karst (Botosaneanu’s Cave, Morrocoy peninsula). These two species are the only representatives of their respective groups in South America and they were the result of the 1982 Amsterdam Expedition to the Venezuelan Islands and other localities on the mainland (Romero, Anartia, 2019). Both species are rejected here as troglobic in Venezuela.
3 REFERENCES
[1] Gallão & Bichuette (ZooKeys, 2018), a primarily Brazilian troglobic list.
[2] White & Culver (Enciclopedia of Caves, 2019, 3th edition), general and generic diversity in caves worldwide.
[3] Freshwater Animal Diversity Assessment (Hydrobiologia, 2008), worldwide.
[4] Deharveng & Bedos (BOOK, chap. 7, 2018), worldwide.
[5] Palacios y Vargas (UMAE, 2013), for Mexico.
[6] Zampulo & Prous (Fauna Cavernicola do Brasil, 2022), for Brazil.
[7] L.F.Oliveira et al. (African Journal of Ecology, 2025), for Africa.
[8] Jugovic, J. et al. (EJT, 2024), for Iran.
[9] Malek-Hosseini & Zamanai (Subterranean Biology, 2017), for Iran.
[10] Malek-Hosseini et al. (Subterranean Biology, 2022), for Iran.
[11] Almanaque Z/South America Cave Smaller Divesities, for Guyana, Suriname, French Guiana, Argentina, Chile, Paraguay and Uruguay.
[12] M. T. Guzik et al. (Invertebrate Systematics, 2010), for W Australia, unique data for this country.
[13] Díaz et al. (Biodiversidad de Cuba, cap. 15, 2015), for Cuba.
[14] Subterranean Fishes of the World (SFW) in December 15, 2024, worldwide.
[15] Furtado et al. (International Journal of Speleology, 2022) for Bolivia.
[16] Campos-Filho et al. (Nauplius, 2023) for Peru.
[17] Galán & Herrera (Boletín de la Sociedad Venezolan, Espeleologia-BSVE, 2006), for Venezuela.
[18] Almanaque Z/Mexican Cave Fauna in December 15, 2024 (SEE), for Mexico.
[19] Angarita-Sierra (CCE/2018 | Chapter Book, 2019) + Moreno-González, J.A. et al. (American Museum Novitates, 2023) for Colombia.
[20] Chatelliers et al. (Freshwater Biology, 2009), for Clitellata worldwide.
[21] Turbanov, I. et al. (PART 1, 2016 | PART 2, 2016), for former Sovietic Union.
[22] Peter Beron (Historia Naturalis Bulgarica, 2015), for SE Asia to New Guinea, including S China and S Japan.
[23] Nicholas, B.G. (The American Midland Naturalist, 1962), for Guatemala to Panama, and Caribbean except Cuba and Jamaica.
[24] Stewart B Peck (Canadian Journal of Zoology, 1999), for Jamaica.
[25] Culver et al. (Conservation Biology, 2021), for USA and Canada.
[26] Martínez, A. et al. (BOOK, 2016), for anchialine caves in Canary Is.
[27] Perez-Moreno, JL et al. (International Journal of Speleology, 2016), for anchialine caves worldwide.
[28] Golovatch, S. et al. (Invertzol, 2018), for European Russia.
[29] Sket et al. (Balkan Biodiversity, 2004), for Balkans in Europe.
[30] Zagmajster, M. et al. (Global Ecology and Biogeography, 2014), for European Pancrustacea.
[31] Pacheco, G. et al. (Insect Conservation and Diversity, 2020), for Guatemala.
[32] Palacios-Vargas et al. (EBM, 2015), detailed checklist from Mexico.
[33] Niemiller, M.L. et al. (Biodiversity and Conservation, 2025), detailed checklist from the USA and Canada.
[34] Hamilton Smith (Australian Journal of Entomology, 1967), for Australia.
4 TAXONOMIC SCOPE
Considering Metazoa as a single group, more than 100 countries worldwide have troglobic representatives. If the goal is to reduce this to two monophyletic groups, the only viable topology is Porifera + Eumetazoa. Under this framework, only Croatia, Mexico, and Brazil have troglobic representatives in both groups. If divided into three groups, within the scope of troglobic fauna, the arrangement would be Porifera + [Cnidaria + Bilateria]. In this case, only Croatia has species in all three groups, making it the country with the greatest phylogenetic breadth of troglobic fauna — followed by Brazil and Mexico in second place, and all other countries in third.
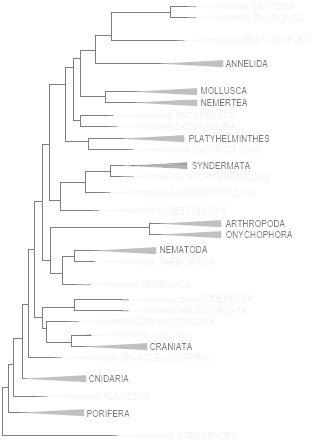
PHYLOGENY OF TROGLOBIC METAZOA (BRAZILIAN METAZOA | PROCEEDINGS OF THE ROYAL SOCIETY B, 2023 | BIODIVERSITY AND CONSERVATION)
5 WORLD NUMBERS
How Many Species of Troglobites [troglobic] Are There? This is the title of an article by Culver & Holsinger (National Speleological Society, 1992), where they estimate there are between 50,000-100,000 troglobic species on the planet. Hobart M. King (Geology) cites a total of 7,700 spp.
We present below numbers from all regions around the world. However, it is important to highlight that these numbers are subject to various biases: many are poorly accurate, do not follow strong criteria regarding the concept of troglobic adopted, some are heavily outdated, much information is unavailable or not compiled, and several conflicts between these numbers can be pointed out. We use them here as reference points for compilation within the most reliable data available. We have extreme consistent data for former URRS (including Central Asia), Africa, Iran, SE Asia to New Guinea and Vanuatu, China, Canada/USA, Mexico, Guatemala to Panama, Caribbean (Jamaica and Cuba separately), Venezuela, Colombia, Ecuador, Peru, Bolivia and Brazil, and few consistent data for Europe (especially in Coleoptera and Diplopoda), Middle East except Iran, India Subcontinent, Japan, Australia, smaller Pacific islands except Vanuatu, New Zealand, Guianas and Cono Sur.
In May 14, 2026 our work believes that 9,499 troglobic documented in world (excluding 4 overlooping species): 393 from Africa and Madagascar, 4,137 troglobic from Europe to Asiatic Russia, 84 in Middle East to Central Asia and Indian Subcontinent, 1,102 in E & SE Asia to New Guinea, 539 in Australia and Pacific Islands, and 3,249 in New World.
TABLE


OVERLAPPING SPECIES
At least 4 species occur in more than one area: Erpobdella absoloni Johansson, 1913 in Western Europe and the former USSR region (Georgia), Prietella phreatophila Carranza, 1954 in the USA and Mexico, Onychiurus acuitlapanensis Palacios-Vargas & Deharveng, 1982 (Onychiuridae) from Venezuela and Guerrero state in Mexico (Galán & Herrera, BSVE, 2006), and Phreatobius sanguijuela Fernandez, Saucedo, Carvajal and Schaefer 2007 from NE Bolivia and WC Brazil.
HOTSPOTS
A hotspots of subterranean biodiversity is a site with 25 or more troglobic, excluding deep soil sites or hyporheic sites unconnected with caves. There are 28 hotspots, that represent almost all subterranean biodiversity hotspots documented thus far at the world scale, in USA (5), Mexico (1), Brazil (3), Bermuda (1), Spain (3), France (4), Slovenia (2), Croatia (1), Bosnia & Hercegovina (1), Romania (1), China (1), Thailand (1), Vietnam (1), Indonesia (1) and Australia (2).
The world has 29 hotspots of troglobic species diversity, defined as cave systems with 20 or more terrestrial or aquatic species (Nǎpǎruş-Aljančič, M et al. BioScience, 2026). The only additional potential hotspots that we are aware of, with 19 known terrestrial subterranean specialists, are Ganxiao Dong, in China (Huang et al. 2021, Diversity), and Fern Cave, in Alabama, in the USA (Diversity). In addition, Moutaouakil and colleagues (2024) reported that Aziza Cave in Morocco has 12 species and an additional 10 (mostly Collembola) are potentially troglobionts but are insufficiently studied to make a definitive statement. Other cave system includes Coume Ouarnède in France (Diversity).
For terrestrial species, two Brazilian caves rank as the 4th and 5th most diverse — Água Clara Cave System and Igatu Cave System (both with 35 spp.) — surpassed only by Vjetrenica (45, Bosnia & Herzegovina), Postojna–Planina Cave System (43, Slovenia), and Cueva del Viento (42, Canary Islands), all located in the European/Macaronesian region.
For aquatic species, Brazil has no hotspots. The highest total recorded for a Brazilian cave is Água Clara Cave System, with only 8 stygobitic species, whereas 20 hotspots worldwide have higher numbers of aquatic species. Only three sites harbor both 20 aquatic specialists and 20 terrestrial specialists: Vjetrenica, Postojna–Planina Cave System, and Križna Jama, all within the Dinaric Karst in Slovenia.
Aquatic hotspots, in descending order of stygobitic species (excluding Copepoda, Ostracoda, and Oligochaeta), are: Walsingham Caves (63, Bermuda, Diversity), Postojna–Planina Cave System (62, Slovenia, Diversity), San Marcos Well (55, USA, Diversity), Vjetrenica (48, Bosnia & Herzegovina, Diversity), Ojo Guareña (46, Spain, Diversity), Robe River Well (43, Australia, Diversity), Cent Fonts (43, France, Diversity), Baget System (40, France, Diversity), Lez Aquifer (39, France), Túnel de la Atlántida (34, Canary Islands), Comal Springs (32, USA), and Križna Jama (31, Slovenia, Diversity).
Terrestrial hotspots are: Vjetrenica (45, Bosnia & Herzegovina, Diversity), Postojna–Planina Cave System (43, Slovenia, Diversity), Cueva del Viento (42, Canary Islands, Diversity), Água Clara Cave System (35, Brazil, Diversity 1 | Diversity 2), Igatu Cave System (35, Brazil, Diversity), Tham Chiang Dao (33, Thailand, Diversity), Mammoth Cave (32, USA, Diversity), Undara Lava Tube System (30, Australia, Diversity), Križna Jama (28, Slovenia, Diversity), Sistema Huautla (27, Mexico, Diversity), Hang Mò So (27, Vietnam, Diversity), Towakkalak System (26, Indonesia, Diversity), Lukina Jama–Trojama (25, Croatia, Diversity), Movile Cave (25, Romania, Diversity), Feihu Dong (23, Guangxi/China, Diversity), Crystal Cave (23, Tennessee/USA, Diversity), and Areias Cave System (22, Brazil).
OTHER NOTES
Only Brazil, Australia and Spain has non karstic hotspots (Culver, Deharveng, Bedos & Pipan, Diversity, 2021). Additionally, a new hotspot from Brazil was described in 2025: Padre Cave, Santana municipality, Bahia state (Community Ecology, 2025). If we consider only the systems with both troglobiont and stygobites, they are reduced to 20: USA (3), Brazil (3), Spain (1), France (3), Slovenia (2), Croatia (1), Bosnia & Hercegovina (1), Romania (1), China (1), Thailand (1), Indonesia (1) and Australia (1). If we consider only the systems with 8 or more troglobic in both groups (hyperhotspots), they are reduced to 13: USA (3), Brazil (1), Spain (1), France (2), Slovenia (2), Croatia (1), Bosnia & Hercegovina (1), Romania (1) and Indonesia (1).
EUROPE EXCEPT RUSSIA (3,852)
Although Europe is by far the region of the world with the greatest diversity of described troglobic forms, continent-wide syntheses by group are rare, and the totals reported for many groups are often based on data from specific regions. [2] mentions 5,000 species, although the unified data compiled here does not support this number. Croatia includes 299 troglobionts and 170 stygobitic taxa, including more 100 beetles (Gottstein, S. et al., Natura Croatica, 2002).
Numbers for Europe except Russia and post-Sovietic state includes (for detailed numbers, check each group's topic):
Porifera (1, Eunapius subterraneus Sket & Velikonja, Croatia)[29],
Cnidaria (1, Velkovrhia enigmatica Matjašić & Sket, 1971, Slovenia, Croatia, and Bosnia & Hercegovina)[29],
Platyhelminthes (112, 99 Tricladida and 13 Temnocephalida)[21][29],
Nemertea (2, Prostoma puteale Beauchamp, 1932, found in France, Switzerland, and Germany, and P. hercegovinense Tarman, 1961, found in Bosnia & Herzegovina)[2],
Nematoda (7, Du Preez, Nematology, 2017, in Slovenia, Belgium, Austria, Hungary, and Romania),
Gastropoda (368, 143 stygobites from France[2], 169 stygobites from the Balkans in Falniowsk, A. et al., Hydrobiologia, 2021, and 56 troglobic[2]),
Bivalvia (1[29], Congeria kusceri Bole, 1962, Slovenia to SW Bosnia and S Dalmatia),
Annelida (103, being Marifugia cavatica Absolon & Hrabe, 1930, known from caves situated in Dinaric Karst in NW Italy, Slovenia, Croatia, and Bosnia & Hercegovina[29], two Sabelliida Nemanereis in Subterranean Biology, 2020, and 100 Clitellata[20]),
Acari (8, Belgium-4, SEE, and Italy-4, SEE | SEE),
Araneae (194, Scientific Data, 2022),
Pseudoscorpiona (132[29], Balkans),
Opiliona (21[29], Balkans),
Palpigrada (23),
Scorpiona (1, Euscorpius studentium Karaman, 2020, Montenegro, Biologia Serbica),
Symphyla (2, Slovenia),
Diplopoda (191, 29 | Iberian Peninsula | Romania),
Chilopoda (55, 50 Lithobiidae[2], Cryptopidae in Spain and Italy, and Geophilidae in France and Croatia),
Ostracoda (114[30]),
Copepoda (547[30]),
Branchiopoda (7[30], Cladocera),
Isopoda (337[30]),
Amphipoda (438[30]),
Bathynellacea (106[30]),
Thermosbaenacea (2[30]),
Mysida (3[30]),
Decapoda (16[30]),
Colembola (338, Fiera, C. et al, JZSER, 2021),
Diplura (110, Sendra et al., Zoological Journal of Linnean Society, 2021),
Coleoptera (607, 384 of them from Western Europe in Balkans[29], 25 in Spain/SEE, 54 in Ariege in France/SEE, 143 in Romania/SEE, 1 in Italy/SEE),
Dermaptera (1, Canary Islands, ZooKeys, 2019),
Zygentoma (1, W Balkans)[29],
Diptera (2, W Balkans)[29],
Hemiptera (4 from Canary Islands, Hoch et al., Subterranean Biology, 2025),
Actinopteri (2, Telestes karsticus Marčić and Mrakovčić 2011 from Croatia, and Barbatula sp. from S Germany)[14], and
Amphibia (1[29], Proteus anguinus Laurenti, 1768), totalling 3,858 troglobic in Europe.
Europe (especially Balkans) shared with Brazil and Mexico the unique troglobic Porifera worldwide (1, in Balkans), shared with Brazil the unique Bivalvia worldwide (1, in Balkans), shared with Brazil and New Zealand the unique troglobic Nemertea (2, France, Switzerland, Germany, Bosnia and Herzegovina), shared with USA the unique troglobic Amphibia (1, Italy, Slovenia, Croatia, and Bosnia-Herzegovina), shared with Colombia (possibly) the unique Nematoda worldwide (7, Belgium, Austria, Hungary, Slovenia, Romania), and has exclusivity among Temnocephalida (15), freshwater troglobic Sabellida (1, Italy, Slovenia, Croatia, Bosnia & Hercegovina) and Cnidaria worldwide (1, Slovenia, Croatia, Bosnia & Hercegovina).
Troglobic Onychophora, Symphyla, and Pauropoda are fully unknown in Europe in any form.
The Canary Islands are the richest volcanic region in the world in subterranean adapted fauna (294 troglobionts and stygobionts), followed by the Hawaiian Islands and the Undara Cave in Australia. Most of the subterranean adapted aquatic fauna from the Canary Islands is restricted to the anchialine environments in La Corona lava tube in Lanzarote, while the oligohaline stygobiont fauna, usually found in groundwater or interstitial freshwaters, is scarcer and represented by a few species of amphipods, copepods, and a single polychaete annelid recorded from Fuerteventura. In fact, Cueva de Felipe Reventón (37) and Cueva del Viento-Sobrado (36), both in Tenerife, are the second and third caves with the greatest number of troglobiont species in the world ranking, while La Corona lava tube, in Lanzarote, is the fourth richest in anchialine species (Nuñez, J., Subterranean Biology, 2020).
AFRICA AND MADAGASCAR (393)
L.F.Oliveira et al. (African Journal of Ecology, 2025)[7] lists 379 troglobic species in Africa.
PLATYHELMINTHES (4) — Tricladida (Dendrocoelum-2, Novomitchellia-1), Neorhabdocoela (1, unknown genus).
MOLLUSCA/GASTROPODA (17) — Atebbania-1, Belgrandiella-1, Bythinella-1, Giustia-7, Heideella-3, Iglica-1, Maroccopsis-1, Neohoratia-1, and Thapsia-1.
ANNELIDA (4) — Astacopsidrilus-1, Metataxis-1, Pristina-1, Trichodrilus-1.
ONYCHOPHORA (1) — Peripatopsis-1 (South Africa).
ACARI/TROMBIFIFORMES (1) — Anomalothrombium-1.
ARANEAE (16) — Acontius-1, Agraecina-1, Andoharano-4, Bathyphantes-1, Blanioonops-1, Cangoderces-1, Dysdera-2, Loxosceles-1, Meta-1, Paraleptoneta-1, Theotima-1, and Troglohyphantes-1.
SCHIZOMIDA (1) — Trithyreus-1 (Tanzania).
PSEUDOSCORPIONA (7) — Botswanoncus-1, Gymnobisium-1, Istriana-1, Neobisium-1, Roncus-2, and Selachochthonius-1.
OPILIONA (2) — Speleomontia-1 and Speleosiro-1.
AMBLYPYGI (1) — Charinus-1.
CHILOPODA (3) — Scolopendromorpha (Cryptops-1, Otostigmus-1) and Lithobiomorpha (Lithobius-1).
DIPLOPODA (4) — Dalodesmidae (Harpethrix-1), Paradoxosomatidae (Jeekelosoma-1), Chordeumatida (Origmatogona-1), and Spirostreptidea (Odontostreptus-1).
SPELAEOGRIPHACEA (1) — Spelaeogriphus-1 (South Africa).
OSTRACODA (4) — Marococandona-2, Meischcandona-1, and Namibcypris-1.
INGOLFIELLIDA (1) — Ingolfiella-1.
AMPHIPODA (81) — Afridiella-1, Austroniphargus-2, Bogidiella-4, Bollegidia-1, Crangonyx-1, Davicookia-1, Dulzura-1, Dussartiella-3, Echinogammarus-2, Eriopisella-2, Gammarus-1, Hadzia-1, Longipodacrangonyx-1, Libertinia-2, Maghrebidiella-3, Metacrangonyx-24, Nubigidiella-1, Paramellita-1, Proleleupia-1, Psammogammarus-1, Pseudoniphargus-7, Reinhardia-1, Ruffohyale-2, Salentinella-1, Sandro-2, Sternophysinx-7, Stygobarnardia-1, Trogloleleupia-5, and Tunisopisa-1.
ISOPODA (84) — Acanthastenasellus-1, Afrocerberus-1, Annina-1, Anopsilana-1, Castellanethes-6, Cirolana-1, Gabunillo-1, Haptolana-1, Johannella-1, Magniezia-6, Marocolana-1, Metastenasellus-7, Microcerberus-1, Microcharon-10, Namibianira-4, Niambia-3, Proasellus-11, Protocerberus-1, Protojanira-2, Protojaniroides-1, Saharolana-1, Skotobaena-2, Stenasellus-7, Thyphlocirolana-10, Trichoniscus-2, and Turcolana-1.
CARIDEA (13) — Caridina-7, Parisia-4, Typhlocaris-1, and Typhlopatsa-1.
THERMOSBAENACEA (5) — Monodella-1, Tethysbaena-3, and Thermosbaena-1 (Tunisia).
BATHYNELLACEA (24) — Acanthobathynella-1, Agnathobathynella-1, Cieniobathynella-1, Cteniobathynella-1, Ctenophallonella-1, Haplophallonella-1, Heterodontobathynella-1, Hexabathynella-5, Iberobathynella-1, Lamtobathynella-1, Nannobathynella-1, Nilobathynella-1, Notobathynella-1, Nunubathynella-1, Paraiberobathynella-1, Parvulobathynella-2, Racovitzaibathynella-2, and Thermobathynella-1.
BRACHYURA (2) — Skelosophusa-2 (Madagascar).
COPEPODA (67) — Copepoda/Calanoidea (Arctodiaptomus-1), Copepoda/Harpacticoida (Elaphoidella-2, Leptocaris-1, Kinnecaris-7, Monodicaris-1, Nitokra-1, Nitocrella-2, Nitocrellopsis-2, Parapseudoleptomesochra-2, Parastenocaris-20, Schizopera-2, Schizoperoides-1), and Copopoda/Cyclopoida (Allocyclops-3, Bryocyclops-4, Haplocyclops-4, Halicyclops-1, Megacyclops-1, Mesocyclops-2, Metacyclops-1, Microcyclops-2, Psammophilocyclops-2, Speocyclops-1, Thermocyclops-3, Typhlamphiascus-1).
COLLEMBOLA (4) — Acherontiella-1, Pseudosinella-1, Troglopedetes-1, and Typhlogastrura-1.
DIPLURA (3) — Anisocampa-1, Jeannelicampa-1, and Tachycampa-1.
HEMIPTERA (1) — Typhlobrixia-1.
DIPTERA (1) — Sergentomyia-1 (Tanzania).
ORTHOPTERA (1) — Spelaeiacris-1.
HYMENOPTERA (1) — Phaenopria-1 (Tanzania).
BLATTARIA (5) — Alluaudellina-1, Nocticola-3, and Typhloblattodes-1.
COLEOPTERA (22) — Antoinella-2, Apteranillus-2, Apteraphaenops-2, Anopsapterus-1, Atheta-1, Brachynillus-1, Domene-3, Duvalius-1, Ifridytes-1, Laemostenus-3, Oedichirus-1, Subilsia-1, Timosmithus-1, Trechus-1, and Typhlozyras-1.
LEPIDOPTERA (1) — Wegneria-1.
DERMAPTERA (1) — Diplatys-1.
ACTINOPTERI (1) — Clarias-1 (Namibia).
36 countries of Africa has troglobic[7], mainly in Morocco (92), Madagascar (64), Algeria (45), South Africa (34) and Namíbia (16).
Addionally, five countries in Africa has 10 troglobic Actinopteri: Cameroon (1:1/1), Somalia (2:3/3), D.R.Congo (1:1/1) and Madagascar (2:2/5)[14]; two troglobic Scorpion is cited for Madagascar — Troglotityobuthus gracilis (Fage, 1946) and Opisthacanthus pauliani Lourenço & Goodman, 2008 (Zoosystema, 2008); and [20] cites Haplotaxis gastrochaetus Yamaguchi (1953) and Villiersia guanivora Omodeo (1987), both Haplotaxidae, from Guinea — 14 added species.
Thus, Africa has 393 troglobic species. For details among Malacostraca, Copepoda and Ostracoda from Africa, see Raoul, T.K. et al (Crustaceana, 2012). Aziza cave in SE Morocco includes remarkable 22 troglobiotic and 4 stygobitic species, mainly in Coleoptera (5), Araneae (4), Entomobryomorpha (3) and Isopoda (2), by S.Moutaouakil et al. (Subterranean Biology, 2024).
FORMER USSR EXCEPT BALTIC COUNTRIES (276)
[21] lists 287 spp. from former USSR except Baltic countries (EX-USSR/SS).
RUSSIA (150, mainly in Crimea and Caucasus)
Tricladida-3, Annelida-2, Gastropoda-9, Bivalvia-1, Opiliones-5, Pseudoscorpiona-3, Acari-5, Chilopoda-1, Diplopoda-5, Ostracoda-4, Copepoda-19, Bathynellacea-6, Amphipoda-22, Isopoda-19, Decapoda-1, Collembola-21, Diplura-6, and Coleoptera-18.
ABKHAZIA (68)
Tricladida-1, Annelida-4, Gastropoda-12, Bivalvia-2, Opiliones-3, Palpigrada-1, Chilopoda-1, Diplopoda-4, Copepoda-5, Amphipoda-12, Isopoda-3, Decapoda-4, Collembola-6, and Coleoptera-10.
GEORGIA (35)
Annelida-3 (includes Erpobdella absoloni Johansson, 1913, also in W Europe), Gastropoda-4, Bivalvia-1, Opiliones-1, Pseudoscorpiona-2, Diplopoda-1, Ostracoda-1, Copepoda-7, Bathynellacea-1, Amphipoda-2, Isopoda-3, Decapoda-2, Collembola-2, Diplura-1, and Coleoptera-4.
UKRAINE (13)
Bathynellacea-3, Gastropoda-1, Amphipoda-1, Collembola-4, Coleoptera-3, and Copepoda-1.
ARMENIA (1)
Bivalvia-1.
AZERBAIJAN (4)
Amphipoda-2 and Collembola-2.
KAZAKHSTAN (2)
Amphipoda-2.
UZBEKHISTAN (5)
Copepoda-1, Annelida-1, Bathynellacea-1, Isopoda-1 and Collembola-1.
KYRGYSTAN (5)
Bathynellacea-4 and Isopoda-1.
TURKMENISTAN (4)
Isopoda-2, Gastropoda-1 and Coleoptera-1.
Belarus, Moldova and Tajikistan does no has troglobic. Five of these species are shared: two Russia/Abkhazia (Conulopolita cavatica/Gastropoda and Nemaspela abchasica/Opiliones), 1 Armenia/Georgia (Euglessa subterranea, Bivalvia), two in Russia/Abkhazia/Georgia (Bryocamptus innominatus and Echinocamptus georgevitchi, both Copepoda). After excluding 7 redundant repetitions (12 repeated entries involving these 5 species), we arrive at 280 validly cited species so far: Tricladida-4, Annelida-10, Gastropoda-26, Bivalvia-4, Opiliones-8, Palpigrada-1, Pseudoscorpiona-5, Acari-5, Chilopoda-2, Diplopoda-10, Ostracoda-5, Copepoda-29, Bathynellacea-15, Amphipoda-41, Isopoda-29, Decapoda-7, Collembola-36, Diplura-7, and Coleoptera-36.
Additionally, one more species troglobic species is listed for Kyrgystan (Chthonius kirghisicus); two more for Turkmenistan in articles: Turkmenocampa mirabilis Sendra & Stoev, 2017 (Diplura), found in Kaptarhana cave, in Lebap province (Sendra, A. et al., Subterranean Biology, 2017), and Paracobitis starostini Parin, 1983 (Actinopteri, Nemacheilidae)[14]; and at least 12 spp. (WHC Unesco) in informal lists: Gammarus troglomorphus, G. parvioculatus, and Bryocyclops jankowskajae as stygobitic Arthropoda; Kainarella minima, Pseudocaspia ljovushkini, P. starostini, P. kainarensis, and Martensamnicola cf. brevicula as stygobitic Gastropoda; and undescribed millipede Bollmania sp., a Pseudoscorpion of family Chernetidae, as well as and spiders Pholcus parthicus and Megalepthyphantes nebulosoides.
Thus, troglobic in EX-USSR/SS are 295, being Tricladida (4), Annelida (10, Erpobdella absoloni Johansson, 1913 shared with W Europa), Gastropoda (31), Bivalvia (4), Acari (5), Pseudoscorpiona (7), Araneae (2), Opiliones (8), Palpigrada (1), Chilopoda (2), Diplopoda (11), Ostracoda (5), Copepoda (30), Bathynellacea (15), Amphipoda (43), Decapoda (7), Isopoda (29), Diplura (8), Collembola (36), Coleoptera (36) and Actinopteri (1).
MIDDLE EAST EXCEPT IRAN PLUS AFGANISTAHN (13)
Middle East and Afganisthan no has unified checklist of troglobic available. At least 13 troglobic was identified for this region, in Scorpiona (1, Israel, Akrav israchanani, SEE), Diplura (1, Sendra et al., Zoological Journal of Linnean Society, 2021, in Lebanon), Annelida/Namanereis (Nereididae, in Oman-1 and Socotra-2) [2], Annelida/Clitellata (2, Oman)[20], and six Actinopteri, in Iraq (2:3/3) and Oman (1:1/3)[14].
IRAN (49)
Iran includes 51 troglobic by [8], being 46 aquatic and five terrestrial. However, we were only able to identify 48 of them in this work, in Amphipoda (24), Copepoda (12), Isopoda (3)[8], Araneae and Diplopoda one each[9], Clitellata (1, Erpobdella borisi Cichocka & Bielecki, 2015), Gastropoda (1), and (2:2/)5 Actinopteri[14]. Addtionally, one Hymenoptera was collected in latter (Yavnella laventa Griebenow, Moradmand & Isaia, 2022, Invertebrate Systematics, 2022).
INDIAN SUBCONTINENT (22)
Indian Subcontinent (Pakistan, India, Bangladesh, Nepal, Butan and Maldivas) no has unified checklist of troglobic available. 21 troglobic Actinopteri are known from this regions, in India (7:10/21)[20], and one Diplura, also in India (Sendra et al., Zoological Journal of Linnean Society, 2021).
JAPAN (90)
Japan no has unified checklist of troglobic available — nearly 200 spp. of troglobic are known from the groundwaters of Japan, most of these troglobic species, 16 of 77 genera, and what is more, 4 of 47 families are endemic to Japan. Uchidastygacaridae, Nipponacaridae, and Kantacaridae are endemic acaridan families of Japan (Matsumoto, Kôichi, International Journal of Speleology, 1976).
Two Clitellata[20], two Staphylinidae (Hlavac et al., Subterranean Biology, 2006), 80 Coleoptera in Shikoku (Ecography, 2006), and one Diplura (Sendra et al., Zoological Journal of Linnean Society, 2021) are troglobic in Japan. (1:1/)5 troglobic Actinoperi, all in Luciogobius (Gobiidae), are known from Japan[14].
KOREA (11)
11 troglobic species are recorded from the Korean Peninsula, all occurring in South Korea: Uenohadesina styx Smetana, 2000 (Staphylinidae, Hlavac et al., Subterranean Biology, 2006), seven Spelaeochthonius (Pseudotyrannochthoniidae, Plos One, 2025), and 3 Pseudocrangonyx (Pseudocrangonyctidae, PeerJ, 2021). Byung-Woo Kim et al. (Korean J. Environ. Biol., 2004) listed 257 cave-dwelling species from the Korean Peninsula — however, the study does not clearly distinguish troglobic from non-troglobic species, which makes it of limited use for assessing the region’s troglobic fauna.
CHINA (724)
We lists 742 troglobic species in China, in Annelida (2, Haplotaxida, Arhynchobdellida), Gastropoda (2, Pulmonata), Araneae (104), Pseudoscorpiona (40), Chilopoda (1), Diplopoda (114), Copepoda (23), Bathynellacea (1, Sinobathynella), Amphipoda (26), Decapoda (23), Isopoda (18), Collembola (42, mainly Coecobrya and Sinella), Diplura (1), Orthoptera (40, all Tachycines), Coleoptera (159), Hemiptera (1), Diptera (2), Actinopteri (Cypriniformes-148, Siluriformes-1) and Anura (1).
[2] cites one cave-dwelling Anura in China, however is fully rejected here as troglobic, being excludes in this count. [14] lists only 120 troglobic Actinoperi in China (data accepted here), being Amblycipitidae-1, Siluridae-1, Balitoridae-3, Cobitidae-8, Cyprinidae-38 and Nemacheilidae-69.
Additionally, five more troglobic are confirmed for China: Annelida/Hirudinea (Novataxa), and Diplura (5, more 4 spp., Sendra et al., Zoological Journal of Linnean Society, 2021).
For these numbers, China includes 716 troglobic species.
SE ASIA FROM MYANMAR TO NEW GUINEA, TIMOR AND PHILIPPINES (280)
[22] lists all the non-Actinopteri troglobionts from SE Asia to New Guinea. Their list contains inconsistencies between the manual counts and the numbers provided in the abstract: 16 fewer species are counted in SE Asia and 6 fewer in New Guinea. Considering only the 11 politically recognized SE Asian countries up to Papua New Guinea (Myanmar, Thailand, Laos, Vietnam, Cambodia, Malaysia, Cingapure, Indonesia, Philippines, Brunei, East Timor, and Papua New Guinea), excluding China and Japan members, we total 146 troglobic species, being
Phyllodocida (1, Namanereis beroni Hartmann-Schröder & Marinov, 1977 from PNG),
Gastropoda (1, Selmistomia beroni Bernasconi, 1995, PNG),
Araneae (13),
Pseudoscorpiones (4),
Scorpiona (2, Troglokhammouanus steineri Lourenço, 2007 in Laos and Vietbocap canhi Lourenço & Pham, 2010 in Vietnam),
Thelyphonida (1, Typopeltis magnificus Haupt, 2004, from Laos),
Opiliones (3),
Acari (4),
Diplopoda (15),
Amphipoda (3, Thailand and Sarawak, all Bogidiellidae),
Isopoda (22),
Decapoda (22),
Diplura (2),
Collembola (23),
Orthoptera (1, Eutachycines cassani (Chopard, 1954), Laos),
Blattaria (8),
Coleoptera (18),
Hymenoptera (1, Leptogenys khammouanensis Roncin & Deharveng, 2003) and
Diptera (2, both Phlebotomus from Philippines).
Additionally, are documented from this regions 37 new troglobic: Chaerilus cavernicola Pocock (Scorpionida, Indonesia, Invertebrate Systematics, 2008), Chaerilus chapmani Vachon & Lourenço (Scorpionida, Malaysia, Invertebrate Systematics, 2008), Orphnaecus pellitus Simon, 1892 (Theraphosidae, Philippines, Subterranean Biology, 2025), two in Diplura (Myanmar-1 and PNG-1, in Sendra et al., Zoological Journal of Linnean Society, 2021), three in Thermosbaenacea (Theosbaena, in Thailand-2 and Cambodia-1, see discussion in Thermosbaenacea topic), and 29 in Actinopteri (Myanmar-1, Thailand-4:5/9, Laos-2:3/3, Vietnan-3:6/7, Malaysia-1, Philippines-2:2/3, Indonesia-3:3/3 and Papua New Guinea-1:1/2)[14]. Two species that were supposedly identified as probable troglobic species in [2] are not recognized as such in [22]: one Temnocephalida and Leiobdella jawarerensis Richardson, 1974 (Clitellate/Haemadipsidae), both for Papua New Guinea.
Additionally, the Towakkalak and Saripa systems in Sulawesi, Indonesia (Diversity, 2021), add 32 species to this initial SE Asian dataset, including species that were mistakenly not highlighted in the text, as well as species that were not previously recognized as troglobic in the original list: Tricladida (2, Dugesia), Acari (Leeuwenhoekiidae-1), Amblypygi (Charinidae-1), Araneae (Ctenidae-1, Pholcidae-2), Opiliones (1), Palpigradi (1, Prokoenenia celebica), Pseudoscorpiones (1), Schizomida (1), Diplopoda (1, Eutrichodesmus reductus), Amphipoda (1, Bogidiellidae), Isopoda (3, Cirolanidae-1, Philosciidae-1, Armadillidae-1), Decapoda/Caridea (Atyidae-4), Decapoda/Brachyura (Gecarcinucidae-1), Collembola (Neanuridae-1, Neelidae-1, Oncopoduridae-1, Sminthuridae-1, Entomobryidae-1), Diplura: (1, Lepidocampa hypogaea), Zygentoma (Nicoletiidae-1), Blattodea (Nocticolidae-2), Coleoptera (Carabidae-1) and Hemiptera (Cixiidae-1).
In turn, Mo So Cave and the Hon Chong Karst of Vietnam (Diversity, 2023) adds 62 species to this list: Gastropoda (Pseudoiglica-1), Acari (Leeuwenhoekiidae-1), Opilioacaridae (Siamacarus-1), Amblypygi (Weygoldtia-1), Araneae (19, Ctenidae-2, Latouchia schwendingeri Decae, 2019, Ochyroceratidae-3, Oonopidae-3, Pholcidae-4, Telemidae-5, Tetrablemmidae-1), Opiliones (3, Epedanidae-2, family undet.-1), Diplopoda (6, Haplodesmidae-1, Pyrgodesmidae-1, Trichopolydesmidae-1, Siphonophoridae-1, Cambalopsidae-2), Amphipoda (Bogidiellidae-1), Isopoda (4, family undet.-1, Sumatrillo-1, Burmoniscus-1, Stenasellus-1), Collembola (15, Hypogastruridae-2, Tullbergiidae-1, Entomobryidae-5, Isotomidae-2, Paronellidae-2, Neelidae-3), Diplura (Japygidae-1), Zygentoma (3), Blattodea (Spelaeoblatta-1), Coleoptera (3, Eustra honchongensis, Curculionidae-1, Pseudochillus honchongensis), and Heteroptera (5, Reduviidae-1, Schizopteridae-1, Cixiidae-2, Delphacidae-1, Kinnaridae-1).
After these additions and considerations, numbers by country/or region: Vietnan (89), Thailand (49), Indonesia/Sulawesi (40), New Guinea (25), Malaysian Sarawak (19), Laos (17), Philippines (15), Sumatra and adjacent Batu (6), Java and adjacent Sumba (6, including one scorpion), Indonesian Kalimatan (4), Myanmar (4), continental Malaysia (4), Cambodia (2), Malaysian Sabah (1), and Moluccas (1).
Thus, SE Asia includes 280 troglobic species.
AUSTRALIA (435)
Australia no has unified checklist of troglobic available, and the lack of precision in the numbers from Australia is absurdly complicating and confusing. Halse (On the Ecology of Australia’s Arid Zone, 2018) presents several inconsistent listings of Australia’s 'subterranean fauna', focusing on strongly positively extrapolated estimates. Some of these figures are at least comparable to the estimated or described troglobic fauna of other regions (e.g., Hemiptera, Diplopoda), whereas others appear highly unrealistic (e.g., Schizomida-59, Diplura-90, Symphyla-38, Annelida-74). These numbers are not considered here, as it is not satisfactorily clear to what extent they actually refer to truly troglobic species.
Our survey on this site, based on direct consultation of freely available online information, allowed us to compile a stable and minimally verifiable list of 435 troglobic species in Australia, within
Gastropoda (4),
Annelida/Phylodocida (1, Namanereis pilbarensis Glasby, Fiege & Van Damme, 2014, Pilbara, Zoological Journal of the Linnean Society, 2014),
Annelida/Phreodrilidae (Astacopsidrilus novus Jackson, 1931)[20],
Acari (EPA-9),
Scopiona (2, Invertebrate Systematics, 2008 | British Arachnological Society, 2001),
Palpigradi (1, Eukoenenia guzikae, Invertebrate Systematics, 2008),
Opiliones (EPA-2),
Araneae (EPA Mesa-1 | Jenolan-2 | EPA-2 | Undara-8),
Pseudoscorpiona (EPA Mesa-2 | Jenolan-1 | EPA-2 | Undara-1),
Schizomida (EPA Mesa-6 | Undara-1),
Chilopoda (4, EPA Mesa-1 | Records of the Western-2 Cryptops, 2006 | Undara-1 Scutigeridae),
Diplopoda (EPA Mesa-2 | Invertebrate Systematics-3 | Undara-4),
Symphyla (1, Hanseniella magna Scheller in Tasmania, Australian Journal of Entomology, 1996),
Ostracoda (74),
Copepoda (83),
Thermosbaenaceae (Halosbaena tulki Poore & Humphreys, 1992 is Western Australia, Wikipedia),
Anaspidacea (Jenolan-1 | EPA-1),
Bathynellaceae (1, EJT),
Amphipoda (140, Diversity, 2024),
Isopoda (EPA-2 | Undara-3),
Caridea (EPA-2, Parisia, Paratya),
Collembola (Jenolan-3 | EPA-2 | Undara-2),
Diplura (EPA Mesa-4 | Records of the Western Australian-1 | Undara-2),
Thysanura (EPA Mesa-1),
Blattodea (9, EPA Mesa-2 | EPA-5 | Undara-2),
Orthoptera (EPA-10),
Heteroptera (EPA-2),
Psocoptera (EPA-2),
Diptera (EPA-4),
Lepidoptera (EPA-1),
Coleoptera (29, EPA-27 | Journal of the Australian Entomological Society-2), and
Actinopteri (3, Synbranchidae-1, Milyeringidae-2, all from Western Australia)[14].
Halse (On the Ecology of Australia’s Arid Zone, 2018) further suggests that Western Australia may represent a potential hotspot exceeding the Balkans, currently regarded as the world’s richest hotspot of subterranean biodiversity, with estimates reaching up to 5,000 troglobionts across its vast semi-arid region. Eberhard & Howarth (Diversity, 2021) reports 19 subterranean biodiversity hotspots in the country, many of which are not included in the filtered figures presented above. If all species cited as troglobic were indeed confirmed as such, and combined with the undetermined hotspot totals—assuming each species to be restricted to a given hotspot — the resulting estimate would reach 733 spp.
In Australia, the terms 'troglofauna' and 'stygofauna' are commonly used, particularly in environmental impact assessments (EIA), to refer to all species collected from terrestrial and aquatic subterranean habitats, respectively. These terms do not distinguish ecological categories, although they are sometimes misinterpreted by non-specialists as equivalent to 'troglobiont' and 'stygobiont'. Regardless of this terminological ambiguity, precise determination of a species’ ecological status is often not critical in most Australian EIA contexts, where the primary focus is on species conservation and extinction risk. The latter is typically assessed based on the sampled and/or inferred distribution range of species in relation to the proposed mining footprint or other areas of potential impact.
Under these conditions, the characterization of Australian troglofauna becomes highly inconsistent and problematic — a truly chaos.
OCEANIA EXCEPT AUSTRALIA AND NEW GUINEA (105)
Subterranean fauna are well documented for New Zealand, where 102 described species are known from groundwater habitats (A Guide to the Groundwater Invertebrates of New Zealand, 2003): Phyllodocida (Namanereis tiriteae (Winterbourn 1969), Clitellata/Phreodrilidae (2)[20], Mollusca (10, all in Hydrobiidae), Acari/Hydracarina (71), Bathynellaceae (5), Anaspidaecea (2, details in Stygocarella pleotelson Schminke, 1980 and Stygocaris townsendi Morimoto, 1977, Beaufortia, 1980), Amphipoda (4), Isopoda (4), and Coleoptera (3).
Additionally, at least one species of troglobic Nemertea is known in New Zealand, Exosphaeroides quirosi Jaume et Queinnec, 2007 (Isopoda) for Vanuatu[22], and one Actinopteri from Guam (Eleotris undescribed species)[14].
Stewart, Robertson & Kasper (NZ Speleological Bulletin, 2023) discuss cave fauna in New Zealand in a highly allegorical manner, which makes the data difficult to use and, therefore, not considered here.
USA/CANADA (1,476)
[33] lists all troglobic from the USA and Canada, which includes Alaska and Hawaii, with (124:288/)1,461 spp., representing six phyla: Platyhelminthes (34), Syndermata (1), Annelida (4:8/22), Gastropoda (39), Nematoda (2), Acari (32), Araneae (121), Opiliones (46), Pseudoscorpiones (153), Schizomida (2), Scorpiones (1), Copepods (18), Ostracoda (22), Bathynellacea (23), Thermosbaenacea (1), Amphipoda (192), Isopoda (118), Decapoda (44), Chilopoda (4), Diplopoda (140), Collembola (101), Diplura (10), Coleoptera (284), Dermaptera (1), Diptera (3), Hemiptera (10), Lepidoptera (1), Orthoptera (6), Zygentoma (8), Actinopterygii (10) and Amphibia (12).
Additionally, Salishbaena kootenai Wagner & Reid, 2021 (Thermosbaenacea, Crustaceana, 2021), two Diplura (EJT, 2026) and 12 more Actinoperi[14] are known as troglobic from region. Thus, USA (including Hawaii and Alaska) and Canada includes 1,476 troglobic.
However, it is important to note that the list for this region includes anchialine forms from Hawaii and parasitic forms in Nematoda, Platyhelminthes, and Acanthocephala — groups that are controversial and/or outside the scope of this text, but which are retained here for the sake of numerical consistency.
Some genera represent remarkable subterranean radiations in the USA, such as Pseudophthalmus (147, Coleoptera), Stygobromus (139, Amphipoda), Pseudotremia (62, Diplopoda), Caecidotea (56, Isopoda), Cicurina (52, Araneae), Tyrannochthonius (36, Pseudoscorpiona), Pygmarrhopalites (31, Collembola), and Batrisodes (30, Coleoptera). Together, these 8 genera account for 553 troglobic species in the USA, representing nearly 2/5 of the national diversity.
Only 11 spp. occur as troglobic in Canada: one Rhagidiidae, three Araneae (Linyphiidae), one Collembola/Entomobryidae, one Collembola/Arrhopalitidae, four Amphipoda/Stygobromus, and one Isopoda/Asellidae. In Hawaii, there are 60: two Rhagidiidae, five Araneae (two Linyphiidae, two Lycosidae, one Ochyroceratidae), five Pseudoscorpiones/Chthoniidae, five Collembola/Entomobryidae, one Collembola/Neanuridae, four Coleoptera/Carabidae, two Diptera, and all troglobic Hemiptera (10), Dermaptera (1), Lepidoptera (1), and Orthoptera (6) from the USA, ten Amphipoda, four Isopoda/Philosciidae, two Atyidae, and two Procarididae. Only two troglobic occur in Alaska: one Amphipoda and one Bathynellacea.
BRAZIL/USA COMPARATION
Neither Brazil nor the USA have troglobic in Cnidaria. Troglobic lineages in Brazil unknown as troglobic in USA are Porifera (2 ✕ 0), Nemertea (3 ✕ 0), Onychophora (1 ✕ 0), Bivalvia (1 ✕ 0), Pauropoda (1 ✕ 0), Symphyla (14 ✕ 0), Blattodea (8 ✕ 0) and Hymenoptera (3 ✕ 0).
Troglobic lineages in USA unknown as troglobic in Brazil are Platyhelminthes/Alloeocoela (1/1), Platyhelminthes/Cestoda (1/1), Platyhelminthes/Trematoda (1/1), Syndermata/Acanthocephala (1/1), Nematoda (2/2), Thermosbaenacea (1/1), Lepidoptera (1/1), and Amphibia (1:2/12).
At commom groups, Brazil and USA are fully tied in Dermaptera,Orthoptera, and Sarcoptiformes.
At commom groups, USA surpasses the Brazil in the following groups (US excedents in parentesis): Coleoptera (3F, ≤19G, 196s), Amphipoda (6F, 19G, 169s), Pseudoscorpiona (2F, ≤20G, 105s), Diplopoda (1G, 46s), Araneae (≤1G, 40s), Decapoda (2F, 6G, 40s), Bathynellacea (1F, 6G, 22s), Ostracoda (1F, 13G, 21s), Trombidiformes (3F, ≤12G, 20s), Tricladida (14s), Copepoda (≤3F, ≤5G, 13s), Annelida (≤2F, ≤6G, 12s), Gastropoda (≤7G, 10s), Opiliones (3s), Mesostigmata (≤1F, ≤1G, 2s), Schizomida (1F, 1G, 1s), Collembola (≤3G), Diplura (≤1G), and Zygentoma (1s).
At commom groups, Brazil surpasses the USA in the following groups at all levels: Chilopoda, Scorpiona, Palpigrada, Hemiptera, Isopoda and Actinopteri.
MEXICO (523)
[5] lists 524 troglobic species in Mexico:
Platyhelminthes (5, Dugesia-3, Opisthobursa-2),
Nematoda (1, Axonchium sbordonii Zullini, 1974),
Annelida/Nereididae (1, Nemanereis cavernicola Solis-Weiss & Espinasa, 1991), Haplotaxida/Acanthodrilidae (Eodrilus-2), Branchiobdellida-Branchiobdellidae (Cambarincola-2),
Gastropoda (8, Amnicolidae-2, Hydrobiidae-3, Lithoglyphidae-3),
Arachnidae/Scorpiones (16, Diplocentridae-4, Euscorpiidae-2, Typhlochactidae-7, Vaejovidae-3),
Arachnidae/Pseudoscorpiona (40, Bochicidae-18, Cheliferidae-1, Chthoniidae-12, Ideoroncidae-7, Syarinidae-2),
Arachnidae/ Schizomida (23, Hubbardidae-14, Protoschizomidae-9),
Arachnidae/Amblypygi (Paraphrynus-6),
Arachnidae/Araneae (58, Agelenidae-2, Cicurinidae-4, Dipluridae-3, Leptonetidae-6, Linyphiidae-1, Nesticidae-4, Ochyroceratidae-2, Oonopidae-1, Pholcidae-25, Tetrablemmidae-1, Theraphosidae-9),
Arachnidae/Opiliones (16, Neogoveidae-1, Phalangodidae-3, Stygnopsidae-11, Nemastomatidae-1),
Arachnidae/Ricinulei (Pseudocellus-11),
Arachnidae/Acari (Prostigmata-2),
Chilopoda (Lithobiidae-2, Scolopocryptopidae-3),
Diplopoda (73, Glomeridesmida-1, Glomerida-6, Spirobolida-1, Spirostreptida-7, Chordeumatida-13, Polydesmida-45),
Copepoda (12, Harpacticoida-1, Calanoida-3, Cyclopoida-8),
Ostracoda (4, Halocyprida-2, Podocopida-2),
Malacostraca/Themosbaenaceae (Tulumella-1),
Malacostraca/Lepidomysidae (Spelaeomysis-3),
Malacostraca/Mysidae (Antromysis-2),
Malacostraca/Stygiomysidae (Stygiomysis-2),
Malacostraca/Amphipoda (22, Bogidiellidae-10, Hadziidae-12),
Malacostraca/Isopoda (52, Anthuridea-2, Flabellifera-23, Microcerberidea-1, Asellota-13, Oniscoidea-13),
Malacostraca/Decapoda (Caridea-21, Procarididea-1, Astacidea-5, Brachyura-5),
Remipedia (Xibalbanus-2),
Collembola (Poduromorpha-11, Entomobryomorpha-27, Symphypleona-4),
Diplura (Campodeidae-4),
Insecta/Zygentoma (Nicoletiidae-13),
Insecta/Orthoptera (Phalangopsidae-6),
Insecta/Hemiptera (Cixiidae-2, Kinnariidae-1),
Insecta/Coleoptera (44, Dytiscidae-1, Carabidae-30, Histeridae-4, Leiodidae-8, Ptinidae-1),
Echinodermata/Asteroidea (1, Copidaster cavernicola Solís-Marín & LaguardaFigueras, 2010), and
Actinopteri (Characidae-1, Ictaluridae-2, Heptapteridae-4, Bythitiidae-1, Synbranchidae-1).
As anchialine species in Mexico in Yucatan peninsula restricted to marine layer of these caves, 16 were excluded from this list: two Remipedia (Xibalbanus, now 3), two Copepoda (Balinella-1, Exumella-1), two Ostracoda (both Halocryptida), two Amphipoda (Bahadzia-2), one Isopoda (Metacirolana mayana (Bowman, 1987)), Procaris mexicana Sternberg & Schotte, 2004, 5 Caridea (Typhlatya dzilamensis Álvarez et al., 2005, Yagerocaris cozumel Kensley, 1988, Triacanthoneus akumalensis Álvarez et al., 2012, Callismata nohochi EscobarBriones et al., 1997, Agostocaris bozanici Kensley, 1988), and the echinoderm Copidaster cavernicola Solís-Marín & LaguardaFigueras, 2010. Additonally, nine new troglobic are adde in checklist of Mexico: one Porifera (1, Legendre et al., Subterranean Biology, 2023, undescribed), Tonalteca spinooculorum (Opiliones, ), five Rhamdia (Heptapteridae, SFW), Caecieleotris morrisi Walsh and Chakrabarty 2016 (Eleotrididae, SFW), and Lucifuga undescribed species 6 (Bythitiidae, SFW). Hlavac et al. (Subterranean Biology, 2006) cite Stenophloeus reddelli Herman, 1969 as troglobic in Mexico; however, it is rejected here[5]. With these changes, the updated list accepted here is as follows, comprising 518 spp.
Porifera (1),
Platyhelminthes (5, Dugesia-3, Opisthobursa-2),
Nematoda (1, Axonchium sbordonii Zullini, 1974),
Annelida/Nereididae (1, Nemanereis cavernicola Solis-Weiss & Espinasa, 1991), Haplotaxida/Acanthodrilidae (Eodrilus-2), Branchiobdellida-Branchiobdellidae (Cambarincola-2),
Gastropoda (8, Amnicolidae-2, Hydrobiidae-3, Lithoglyphidae-3),
Arachnidae/Scorpiones (16, Diplocentridae-4, Euscorpiidae-2, Typhlochactidae-7, Vaejovidae-3),
Arachnidae/Pseudoscorpiona (40, Bochicidae-18, Cheliferidae-1, Chthoniidae-12, Ideoroncidae-7, Syarinidae-2),
Arachnidae/ Schizomida (23, Hubbardidae-14, Protoschizomidae-9),
Arachnidae/Amblypygi (Paraphrynus-6),
Arachnidae/Araneae (58, Agelenidae-2, Cicurinidae-4, Dipluridae-3, Leptonetidae-6, Linyphiidae-1, Nesticidae-4, Ochyroceratidae-2, Oonopidae-1, Pholcidae-25, Tetrablemmidae-1, Theraphosidae-9),
Arachnidae/Opiliones (16, Neogoveidae-1, Phalangodidae-3, Stygnopsidae-12, Nemastomatidae-1),
Arachnidae/Ricinulei (Pseudocellus-11),
Arachnidae/Acari (Prostigmata-2),
Chilopoda (Lithobiidae-2, Scolopocryptopidae-3),
Diplopoda (73, Glomeridesmida-1, Glomerida-6, Spirobolida-1, Spirostreptida-7, Chordeumatida-13, Polydesmida-45),
Copepoda (10, Harpacticoida-1, Calanoida-1, Cyclopoida-8),
Ostracoda (Podocopida-2),
Malacostraca/Themosbaenaceae (Tulumella-1),
Malacostraca/Lepidomysidae (Spelaeomysis-3),
Malacostraca/Mysidae (Antromysis-2),
Malacostraca/Stygiomysidae (Stygiomysis-2),
Malacostraca/Amphipoda (20, Bogidiellidae-10, Hadziidae-11),
Malacostraca/Isopoda (52, Anthuridea-2, Flabellifera-22, Microcerberidea-1, Asellota-13, Oniscoidea-13),
Malacostraca/Decapoda (Caridea-16, Astacidea-5, Brachyura-5),
Remipedia (Xibalbanus-2),
Collembola (Poduromorpha-11, Entomobryomorpha-27, Symphypleona-4),
Diplura (Campodeidae-4),
Insecta/Zygentoma (Nicoletiidae-13),
Insecta/Orthoptera (Phalangopsidae-6),
Insecta/Hemiptera (Cixiidae-2, Kinnariidae-1),
Insecta/Coleoptera (44, Dytiscidae-1, Carabidae-30, Histeridae-4, Leiodidae-8, Ptinidae-1),
Echinodermata/Asteroidea (1, Copidaster cavernicola Solís-Marín & LaguardaFigueras, 2010), and
Actinopteri (11, Characidae-1, Ictaluridae-2, Heptapteridae-9, Bythitiidae-2, Eleotrididae-1, Synbranchidae-1).
Mexico no has described troglobic in Nemertea, Onychophora, Mollusca/Bivalvia, Bathynellacea, Spelaeogriphacea, Blattodea, Dermaptera, Diptera, Hymenoptera, Pauropoda, Symphyla, Palpigradi or Amphibia. When considering species not yet described in Brazil, the country surpasses Mexico in all groups except Nematoda (unknown in Brazil), Schizomida, Ricinulei (unknown in Brazil), Scorpionida, Mysids overall (unknown in Brazil), Ostracoda, Copepoda, Decapoda, and Zygentoma.
AMERICA CENTRAL FROM GUATEMALA TO PANAMA (21)
The only consistent reference on the troglobic fauna of southern Mexico, America Central, and the Caribbean is [23], who lists 170 spp. for the vast region. Currently, Mexico, Cuba, and Jamaica have more recent references and are treated independently. Therefore, we use these references only for the region from Guatemala to Panama and the West Indies, excluding Cuba and Jamaica.
Based on [23], only eight troglobic occur from Guatemala to Panama, six valid nowdays (exclusion of Pselliodes harveyi Chamberlin, 1942 from Panama and Pselliophora cavincola Chamberlin, 1918 from Trinidad, both under Sphendononema guildingii Newport, 1845, Chilobase), namely:
Trombicula cavernarum Ewing, 1933 and
T. trifurca Ewing, 1933 (Trombidiformes, Panama),
Otostigmus cooperi Chamberlin, 1942 (Chilopoda, Panama),
Calymmodesmus inquinatus Causey, 1960 (Diplopoda, Guatemala),
Lepidocyrtus usitatus Folsom, 1927 (Collembola, Panama), and
Ptomaphagus giaquintoi Jeannel, 1936 (Coleoptera, Alta Verapaz, Guatemala).
Additonally, 15 troglobic are added in region, including three crabs in the genus Typhlopseudothelphusa (Pseudothelphusidae, two in Guatemala and one in Belize, Hobbs, Texas Memorial Museum Speleological Monographs, 1986), two Diplura in Guatemala (Sendra et al., EJT, 2026), Rhamdia typhla Greenfield, Greenfield, and Woods, 1982 (Actinopteri/Heptapteridae, Belize)[14], and nine from Gruta de Lanquín in Guatemala[31] (Isopoda-2, Platyhelminthes-2, Araneae-1, Pseudoscorpiones-1, Opiliones-1, Polydesmida-1, and Zygentoma-1).
For these numbers, America Central has 21 documented troglobic. S.Deleva et al. (Subterranean Biology, 2023) discusses the cavernicolous fauna of Costa Rica and confirms that there are no troglobic species in the country, only strong candidates are Pseudothelphusa puntarenas Hobbs 1991 (Decapoda), various specimens of Trogolaphysa (Collembola) and some Acari (Rhagidiidae) from Barra Honda.
CUBA (130)
[13] cites 112 troglobic in Cuba, all Arthropoda: Acari (12), Scorpiones (1), Pseudoscorpiones (3), Amblypygi (1), Schizomida (5), Ricinulei (4), Opiliones (4), Araneae (7), Chilopoda (2), Ostracoda (5), Copepoda (18), Mysidacea (5), Isopoda (7), Decapoda (13), Amphipoda (3), Remipedia (1), Zygentoma (3), Collembola (7), Orthoptera (4), Blattoidea (1) and Coleoptera (6) — here Remipedia (1) is excluded due to anchialine status.
Additionally 20 species are reported as troglobic in Cuba:
Eukoenenia orghidani (Palpigradi, The Journal of Arachnology, 2010),
Alayotityus delacruzi Armas, 1973, Tityopsis sheylae Teruel & Rodríguez-Cabrera, 2020 (Scorpionida/Buthidae, Euscorpius, 2021),
Tethysbaena vinabayesi Wagner, 1994 (Thermosbaenacea, see discussion in Thermosbaenacea topic),
Clitellio cavernicolus Botea, 1983 (Tubificidae)[20], four species of Pristina (Tubificidae)[20],
two Cryptops (Chilopoda, ZooKeys), and
nine Lucifuga (Bythitidae, Ophidiiformes, Actinopteri)[14].
Thus, Cuba has 131 troglobic species in our survey.
JAMAICA (42)
Troglobic in Jamaica contains 43 spp. troglobic[24], being 27 of troglobic in Onychophora (Speleoperipatus spelaeus Peck, 1975), Araneae (9), Opiliones (2), Pseudoscorpionida (4), Schizomida (1), Isopoda (1), Collembola (2), Blattodea (1), Hemiptera (2) and Coleoptera (4), and 16 stygobitic, in Tricladida (1), Ostracoda (3), Mysida (2), Thermosbaenacea (1), Decapoda (2), Amphipoda (4) and Isopoda (3).
Thermosbaenacea record is excluded here due to inconclusive status relative to the literature — thus, we accepted 42 troglobic in Jamaica.
CARIBBEAN EXCEPT CUBA AND JAMAICA PLUS BERMUDA (27)
The only consistent reference on the troglobic fauna of southern Mexico, America Central, and the Caribbean is [23], who lists 170 spp. for the vast region. Currently, Mexico, Cuba, and Jamaica have more recent references and are treated independently. Therefore, we use these references only for the region from Guatemala to Panama and the West Indies, excluding Cuba and Jamaica.
[23] lists nine troglobic for this area, namely:
Maymena bruneti Gertsch, 1960 (Araneae, Trinidad),
Bogidiella bredini Shoemaker, 1959 (Amphipoda, Barbuda),
Metaniphargus beattyi Shoemaker, 1942 (Amphipoda, Virgin Islands),
M. nicholsoni Shoemaker, 1959 (Amphipoda, Barbuda),
Typhlatya monae Chace, 1954 (Decapoda Caridea, Puerto Rico),
Stygiomysis holthuisi Gordon, 1960 (Mysida, Saint Marteen),
Calycuoniscus spinosus Collinge, 1917 (Isopoda, Trinidad),
Porcellionides minutissimus Van Name, 1936 (Isopoda, Bahamas), and
Philoscia bermudensis Dahl, 1892 (Isopoda, Walsingham Cave, Bermuda).
Additionally, 13 more troglobic are confirmed for this area:
Pseudochthonius arubensis Wagenaar-Hummelinck (Aruba, Zoologia, 2023),
Heteronebo clareae Armas, 2001 in Bahamas (Scorpiona, Euscorpius, 2021),
Cazierius ciguayo Teruel, Jiménez & Santos, 2021 and C. cayacoa Teruel, Jiménez & Santos, 2021 in Dominican Republic (Scorpiona, Euscorpius, 2021),
Newportia stoevi Schileyko, 2013 (Chilopoda, Puerto Rico, ZooKeys, 2013),
six Namanereis (Annelida, Nereididae, one shared with Mexico),
Phallodriloides macmasterae Erséus, 1986 (Tubificae) in Bermuda[20], Dero haitiensis Dumnicka, 1986 and Spirospermoides stocki Dumnicka, 1983 (both Tubificidae) in Haiti and adjacent islands[20],
Tethysbaena colubrae (Thermosbaenacea, Puerto Rico),
Rhamdia urichi Mees, 1974 (Heptapteridae, from Trinidad and Tobago)[14], amd two Lucifuga (Bythitidae, Bahamas)[14].
Thus, in West Indies except Jamaica and Cuba are known 22 troglobics.
GUIANAS (0)
[11] cites possibly Lepidocampa (Diplura), Eukoenenia (Palpigrada) and Modisimus (Araneae) as troglobic in French Guiana, however their troglobic status in unknown, and all are rejected here.
VENEZUELA (47)
[17] lists 46 troglobic in Venezuela, in Acari (4:9), Opiliones (4, all in Trinella, Agoristenidae), Amblypygi (2, both Charinus), Scorpionida (1, Taurepania trezzii, Chactidae), Diplopoda (1, Trichopolydesmidae), Collembola (1, Onychiuridae), Copepoda/Cyclopidae (1), Ostracoda (2, in Cyprididae), Bathynellaceae (1, Parabathynellidae), six Isopoda (6:6), Amphipoda (3:3), Decapoda (2, Pseudothelphusidae), Orthoptera (2, both Rhaphidiophoridae), Blattodea (1, Paranocticolla venezuelana), Coleoptera (3:3), and seven fishes (in 4 families).
From this list, we excluded the two anchialine species — Stygocyathura univam (Botosaneanu, 1983)(Isopoda) and Metaniphargus venezuelanus (Stock & Botosaneanu, 1983)(Amphipoda), as they fall outside the scope of this article.
Additionaly, troglobic in Venezuela includes Speleopsocus chimanta Lienhard, 2010 (Psocoptera, Revue suisse de Zoologie, 2015), Krenedrilus papillatus Dumnicka, 1983 (Annelida/Tubificidae)[20], and Cryptocellus armasi Botero-Trujillo and Prendini, 2025 (Ricinulei, Zootaxa, 2025).
Thus, 47 troglobic are recorded in Venezuela.
COLOMBIA (42)
65 troglobic are listed in Colombia[19], in Nematoda (1, Rhabditida), Annelida (2, Haplotaxide, Enchytraeidae one each), Tityus grottoedensis (Scorpionida), Opiliones (7:7), Amblypygi (2, Phrynidae and Paracharontidae), Araneae (16, in 11 families, Barychelidae, Araneidae, Anyphaenidae, Lycosidae, Tetragnathidae, Pisauridae without troglobic in Brazil), Chilopoda (3, undescribed Scolocryptops and 2 Psilliodes), Diplopoda (7), Isopoda (1, Armadillidium), Coleoptera (5, two in Staphylinidae; Carabidae, Nitidulidae, Passalidae one each), Ephemeroptera (2, if confirmed, will be the first troglobis in this order worldwide), Blattoidea (1, Blaberidae), Dermaptera (1), Diptera (2, Drosophilidae and Mycetoohilidae one each), Hemiptera (3, Cydnidae, Cavernicola pilosa in Reduviidae, Strudivelia cinctipes in Veliidae), Lepidoptera (1), Trichoptera (2, in Hydrobiosidae and Hydropsychidae), Actinopteri (7, all Trichomycterus) and Avialia (Steatornis caripenis).
Steatornis caripenis assignated in work as troglobic is a obviously a huge error. Troglobic status rejecteds for both Annelida (Angarita-Sierra et al, Chapter Book, 2019), all Psilliodes (fully rejected by many sources), 13 Araneae (Angarita-Sierra et al, Chapter Book, 2019), Tityus grottoedensis (by original description, Zootaxa, 2014), 4 Opiliones (Angarita-Sierra et al, Chapter Book, 2019), one Ephemeroptera (Angarita-Sierra et al, Chapter Book, 2019), Cavernicola pilosa (Dissertation/FOC) and Strudvelia cinctipes (Biota Colombiana).
Additionally, 10 troglobic Actinopteri are recorded in Colombia (all in Trichomycterus)[14], and Diploexochus cacique (Isopoda) was described in 2025 (ZooKeys).
With these exceptions, Colombia has, with some consistency, 42 spp. of troglobic: Nematoda (1, Rhabditida), Opiliones (3), Amblypygi (2, Phrynidae and Paracharontidae), Araneae (3), Chilopoda (1, undescribed Scolocryptops), Diplopoda (6, in Chelodesmidae, Pyrodesmidae, Spirostreptidae, Stemmiulidae and Rhinocricidae), Isopoda (2, both in Armadillidae), Coleoptera (5, two in Staphylinidae; Carabidae, Nitidulidae, Passalidae one each), Ephemeroptera (1), Blattoidea (1, Blaberidae), Dermaptera (1), Diptera (2, Drosophilidae and Mycetoohilidae one each), Hemiptera (1), Lepidoptera (1), Trichoptera (2, in Hydrobiosidae and Hydropsychidae) and Actinopteri (10, all Trichomycterus) — however, troglobic status of Nematoda, Ephemeroptera, Dermaptera, both Diptera, Lepidoptera and both Trichoptera need strong validation to be fully credited.
ECUADOR (8)
Ecuador no has unified checklist of troglobic available. Literaure lists 8 troglobic species in country: Marionina ecuadoriensis Righi and Hemienchytraeus mauriliae Righi (Clitellata/Enchytraeidae)[20], also Righi (Papéis A vulsos de Zoologia, 1981), Tayos ashmolei Reddell & Cokendolpher, 1984 (Schizomida, British Arachnological Society, 1984) and Troglotayosicus vachoni Lourenço, 1981 (Scorpiona/Troglotayosicidae) from Cueva de los Tayos, Santiago-Morona Province; Ischnoptera peckorum Roth, 1988 (Ectobiidae, Blattaria, Galapagos Species Database, Bellavista Cave) and Pinostygus galapagoensis Cambell & Peck, 1989 (Staphylinidae, Hlavac et al., Subterranean Biology, 2006) from Galapagos islands; and two Actinopteri, Astroblepus pholeter Collette, 1962, and one Chaetostoma undescribed species[14].
PERU (3)
Only three spp. in Peru the following can be considered troglobic[16]: Acheroxenylla lipsae Palacios-Vargas, 2020 (Collembola, Poduromorpha, Hypogastruridae), Astroblepus riberae Cardona and Guerao, 1994 (Actinopteri, Siluriformes, Astroblepidae) and Caecopactes chullachaqui Campos-Filho, Sfenthourakis and Bichuette, 2024 (Isopoda, Scleropactidae).
BOLIVIA (8)
Bolivia no has unified checklist of troglobic available. Six troglobic are cited for Bolivia sampled in Umajalanta cave[15]: Coleoptera (3, one described, Escolatrechus bolivianus Mateu, 2002), Acari (1, Rhagidiidae), Collembola (1, Entomobryomorpha), and Isopoda (1, Dubioniscidae). Additionally, Trichomycterus chaberti Durand 1968 and Phreatobius sanguijuela Fernandez, Saucedo, Carvajal and Schaefer 2007 are troglobic in Bolivia[14], totalling 8 troglobic species in country.
ARGENTINA (31)
Without a unified checklist of troglobic available, our survey counted 31 troglobic in Argentina:
■ 24 known in Cuchillo Curá cave system, largest known cave system in country (ICS/vol VII, 2025), being 21 terrestrial and three aquatic, all Arthropoda, in Acari (1), Pseudoscorpiona/Chthoniidae (1), Opiliones/Laniatores (1), Palpigradi (2), Araneae (3), Symphyla (1), Isopoda (4), Amphipoda (2), Collembola (3), Diplura (3, Campodeidae, Japygidae), Coleoptera/Leiodidae (1), Auchenorrhyncha/Cixiidae (1), and Zygentoma/Nicoletiidae (1) — only six of these species are described: Picunchenops spelaeus Maury, 1988 (Opiliones/Laniatores), Pseudothiphistes pallidus (Millidge, 1991) - a Araneae/Linyphiidae, Notolathrus sensitivus Remes Lenicov, 1992 (Cixiidae), Adelopsis picunche Gnaspini, 1991 (Coleoptera/Leiodidae), Grossogidiella mauryi (Grosso & Fernández, 1993) and Patagongidiella danieli Grosso & Fernández, 1993), both Amphipoda/Bogidiellidae.
■7 in other caves: Troglospinotheca (1, Collembola, Neuquen region, Mémoires de Biospéologie, 1999), four Anaspidacea (Plazi | Plazi), Pleonaraius spelaeus (Diplopoda/Polydesmida, Neuquen, Romero-Rincon & Ferreira, Arthropoda, 2025), and one Actinopteri, Silvinichthys bortayro Fernandez and De Pinna 2005, in Trichomycteridae[14].
PARAGUAY, URUGUAY AND CHILE (2)
For Paraguay, Uruguay and Chile, only two troglobic are documented: two Anaspidacea (Plazi | Plazi), both from Chile.
6 BRAZILIAN NUMBERS AND TAXA
This list of cave-obligate taxa is in a continual state of flux, as new species are added through new species descriptions, some are removed by being synonymized with other species, and others are reclassified as troglophiles or stygophiles when records from surface habitats or other new evidence is obtained. Moreover, we recognize that the inclusion or exclusion of particular taxa may be contentious for some biospeleologists.
NOTES
It is noteworthy that, among the 14 troglobic species recorded from more than one state, 6 belong to Pseudoscorpiones, and several of them exhibit highly disjunct distributions that are unusual for troglobic species. This pattern raises questions about their true troglobic status or whether they may in fact represent multiple morphologically very similar species with the geographically restricted distributions typically expected for troglobic (Prado & Ferreira, Zoological Studies, 2024 — Pseudochtonius strinatii maybe three species!). For example, Spelaeochernes eleonorae has been reported n caves of Mato Grosso do Sul, Goiás, Minas Gerais and São Paulo state.
Pseudonannolene canastra (Diplopoda), a troglobic species, was long regarded as a valid species but is currently synonymized under P. ambuatinga (Iniesta, Bouzan & Brescovit, European Journal of Taxonomy, 2023).
Some species with strong potential to be troglobic were shown, upon their formal description, not to possess this status. For example, Charinus carajas Giupponi & Miranda, 2016 and C. orientalis Giupponi & Miranda, 2016, cited by Zampulo & Prous (Fauna Cavernícola do Brasil, 2022) for Brazil, as well as Tyrannochthonius iuiu and T. aratu (Guimarães, L et al., Zootaxa, 2025), do not meet the criteria for troglobic species.
Charinus giganteus (Amblypygi, São Gabriel, Bahia, Souza & Ferreira, Zootaxa, 2025),
Pseudochthonius itakuatiara (Pseudoscorpiona, Zoological Studies, 2024),
Geogarypus gollumi and Progarypus smaugi (Pseudoscorpiona, Geogarypidae and Hesperolpiidae, respectively, Bedoya-Roqueme et al., Zootaxa, 2022),
Paracymbiomma pepita, P. otxurucu and P. una (Araneae, Cizauskas, I. et al., Zootaxa, 2024),
and
Whiteheadiana thaisae (Carabidae, Benumea et al., Zootaxa, 2023), may be troglobic; however, the present study is unable to confirm their troglobic status.
Would Kadiweuoniscus rebellis be the species referred to as Oniscoidea 1, in Cordeiro et al. (Biota Neotropica, 2014)? No indication denying or affirming this was found in our research, thus here we consider them as distinct taxa.
TABLE


DETAILED NUMBERS
The 893 troglobic species recorded in Brazil include both described and undescribed taxa, as well as species that are either endemic to a single state or more widely distributed. Below, we provide a more detailed breakdown of each category. 376 spp. are described (234 described latter [1], or after but non cited until to date), being 362 state endemic and 14 in two or more states; and 517 remains undescribed, all state endemic.
DESCRIBED IN [1]
[1] cites 150 spp., but 9 are rejected here as troglobits: Troglorhopalurus lacrau (BA, Scorpionida)1, Isoctenus corymbus (Ctenidae)2, Lygromma ybyguara (Prodidomidae)3, and Leonardossia hassalli (PA, Isopoda)4, rejected by [6]; Metagonia potiguar (RN, Araneae)5, non cited at Bento et al. (Biodiversity and Conservation, 2022); Ochyrocera ibitipoca (MG, Araneae)6, rejected in A. Brescovit & I. Cizauskas (Arachnology, 2018); Endecous apterus7 and E. peruassuensis8 (rejected in Carvalho, P.H.M. et al, Zootaxa, 2023); and Ancistrus cryptophthalmus9 (due to epigean populations, Alves, BVA et al, International Journal of Speleology, 2013). Discocyrtus pedrosoi (BA, Opiliones), present in [1] and rejected in [6], is accepted here. Thus, [1] contributes 142 species to the new survey.
DESCRIBED AFTER [1]
A total of 235 troglobic species have been described in Brazil after [1] and are listed below by taxonomic group, with appropriate references.
PORIFERA (1)
Arinosaster patriciae (MT, Neotropical Biology and Conservation, 2021).
PLATHELMINTHES (8)
Sluysia triapertura (RN, Invertebrates Systematics, 2018), Girardia arenicola, G. corumbataiensis, G. nobresis, G. paucipunctata, G. spelaea (SP, MS, BA, Zoologischer Anzeiger, 2021), Girardia patiensis, and G. alba (BA, Zoologischer Anzeiger, 2025).
MOLLUSCA/BIVALVIA (1)
Eupera troglobia (TO, Subterranean Biology, 2022).
MOLLUSCA/GASTROPODA (6)
Gastrocopta sharae (GO, Zoosystematic Evolution, 2017), Habeastrum parafusum, H. omphalium (MS, Zootaxa, 2019), Spiripockia umbraticola (BA, Journal of Natural History, 2021), Idiopyrgus eowynae and I. meriadoci (BA, Zoosystematics and Evolution, 2024).
ARTHROPODA/ARACHNIDA/SCORPONES (2)
Troglorhopalurus iuiu (BA, Arthropoda, 2025), T. araras (BA, Journal of Natural History, 2026).
ARTHROPODA/ARACHNIDA/TROMBIDIFORMES (1)
Leptus sidorchukae (MG, Zootaxa, 2019).
ARTHROPODA/ARACHNIDA/PSEUDOSCORPIONA (25)
Pseudochthonius ramalho, P. olegario, Spelaeochernes altamirae, S. armatus, S. bahiensis, S. dentatus, S. dubius, S. eleonorae, S. gracilipalpu, S. pedroi, S. popeye (BA, MG, SP, GO, MS, MG, BA, SE, PR, SC, Arachnology, 2022 + Revue Suisse de Zooologie, 2001),
Spelaeobochicha goliath (MG, Zootaxa, 2018),
S. mahnerti (MG, Zootaxa, 2020),
Pseudochthonius koinopoliteia, P. diamachi, P. pali (Prado & Ferreira, Zootaxa, 2023),
Pseudochthonius lubueno (BA, Zoologia, 2023),
Pseudochthonius aware (BA, Zoological Studies, 2024),
Pseudochthonius limettioides (MG, Zootaxa, 2024),
Pseudochthonius maquinensis, P. urubuquaqua (MG, Zootaxa, 2025),
Pseudochthonius cesarioensis, P. gonssaloensis (BA, Zootaxa, 2025),
Ceriochernes spelaeofoliaceus and C. foliaceospeluncarum (BA, MG, Zootaxa, 2025).
ARTHROPODA/ARACHNIDA/PALPIGRADI (14)
Eukoenenia eywa, E. navi, E. neyriti (MG, Invertebrate Systematics, 2018),
E. igrejinha (MG, Journal of Arachnology, 2019),
E. ibitipoca (MG, Zootaxa, 2019),
E. jequitai, E. lundi, E. magna (MG, Zootaxa, 2020),
E. audax (GO, Zootaxa, 2020),
Allokoenenia canhembora, A. stygia (BA, PA, European Journal of Taxonomy, 2022),
E. mocororo (MG, Zootaxa, 2022),
E. piloi and E. renatae (MG, Zoological Journal of the Linnean Society, 2025).
ARTHROPODA/ARACHNIDA/AMBLYPYGI (5)
Charinus cearensis, C. diamantinus, C. euclidesi, C. puri and C. renneri (many states, EJT, 2021).
ARTHROPODA/ARACHNIDA/OPILIONES (4)
Iandumoema cuca, I. gollum, I. stygi (MG, Invertebrate Systematics, 2020) and
Spaeleoleptes gimli (BA, EJT, 2024).
ARTHROPODA/ARACHNIDA/ARANEAE (30)
Brasilomma enigmatica (MG, Zootaxa, 2012),
Tonton itabirito, T. matodentro (MG, ZooKeys, 2013),
Carajas paraua, Tisentnops mineiro, T. onix (PA, MG, ZooKeys, 2016),
Paracymbiomma pseudocaesus (PA, Dissertation, 2017),
Loxoscelis troglobia (BA, Zootaxa, 2018),
Paracymbiomma caecus (PA, Zootaxa, 2018),
Paracymbiomma bocaina (PA, Zootaxa, 2018),
six Matta (MG, Zootaxa, 2019),
Indiani gaspar (MG, Zootaxa, 2020),
Ochyrocera ritxoco, O. ritxoo (PA, Zookeys, 2021),
Ochyrocera dorinha, O. rosinha, O. ungoliant (MG, PA, Subterranean Biology , 2021),
Speocera babau, S. pinima, S. piquira (PA, Zootaxa, 2022),
Ctenus igatu (BA, Journal of Aracnology, 2022),
Pinelema elinae (BA, Zootaxa, 2024),
Loxosceles bodoquena (MS, EJT, 2024),
Loxosceles boqueirao (BA, EJT, 2024),
and
Paleotoca diminas (MG, Taxonomy, 2024).
ARTHROPODA/CHILOPODA (4)
Schendylops janelao (MG, Zootaxa, 2019), Plutogeophilus jurupariquibaba (SP, Organisms Diversity & Evolution, 2023), Cryptops didi (SP, Journal of Natural History, 2024), and Newportia eleonorae (MS, Zoologischer Anzeiger, 2025).
ARTHROPODA/DIPLOPODA (6)
Dobrodesmus mirabilis (BA, Zootaxa, 2016), Phaneromerium troglopterygotum (BA, Zootaxa, 2022), Strongylosomides troglobius (BA, Zootaxa, 2022), Onciurosoma troglobium (BA, Arthropoda Selecta, 2022), Cayenniola albaserrata (BA, Zootaxa, 2024) and Pandirodesmus zogbiea (PA, Zootaxa, 2025).
ARTHROPODA/MALACOSTRACA/AMPHIPODA (8)
Spelaeogammarus ginae (BA, Nauplius, 2022), S. rafaelae, S. lundi (BA/MG, Taxonomy, 2025), Spelaeogammarus quilombola (BA, Zoological Studies, 2025), Hyalella karajas, H. amazonica, H. nebula, H. uva (PA/PA/MG/RS, Zootaxa, 2025).
ARTHROPODA/MALACOSTRACA/ISOPODA (65)
Pectenoniscus lilieae, Benthana xiquinhoi (BA, Zootaxa, 2019),
Amazoniscus spica, Alboscia jotajota, Metaprosekia igatuensis (PA, SP, BA, European Journal of Taxonomy, 2020),
Xangoniscus aganju, X. lundi, X. dagua, X. santinhoi, X. ceci (BA, MG, Zootaxa, 2020),
Pectenoniscus montalvaniensis, P. juveniliensis, P. iuiuensis, P. carinhanhensis, Pectenoniscus santanensis, P. morrensis (BA, MG, Nauplius, 2020),
Chaimowiczia tatus, C. uai (BA, MG, Subterranean Biology, 2021),
Pectenoniscus fervens, P. pankaru (PI, BA, ZooKeys, 2022),
Spelunconiscus septemlacuum (MG, Nauplius, 2022),
Xangoniscus lapaensis, X. loboi (BA, Subterranean Biology, 2022),
Chaimowiczia obybytyra (BA, Subterranean Biology, 2022),
Iansaoniscus leilae, I. paulae (BA, Subterranean Biology, 2022),
Cylindroniscus platoi (MG, Zootaxa, 2022),
Benthana alba, Benthanoides amazonicus, B. tarzan (PA, Zootaxa, 2023),
Pectenoniscus monsviridis, P. revelatus, P. archaeos, P. sepultus, P. caesarius (BA, Studies on Neotropical Fauna and Environment, 2023),
Caraiboscia jabutiensis, Gabunillo enfurnado, Venezillo moreirai, V. limai (many states, Zoosystema, 2023),
Trichorhina baiana (BA, Biota Neotropica, 2023),
Kadiweuoniscus rebellis (MS, ZooKeys, 2024),
Circoniscus mendesi, C. xikrin (PA, EJT, 2024),
Ctenorillo iuiuensis (BA, Zoosystema, 2024),
Xangoniscus paiabare, X. tymaopeba, X. ykanhema, X. puku (BA, Tropical Zoology, 2024),
Xangoniscus antiquus, X. chaimowiczi, X. jonasi (BA, Subterranean Biology, 2024),
Oiticicarcinia epikarstica, Itararecarcinia yapyra, Poicarcinia jandairensis (RN, Tropical Zoology, 2025),
Circoniscus paradisus (PA, Nauplius, 2025),
Brasileirinho sergipanus (SE, Zootaxa, 2025),
Brasilana spelaea (RN/CE, Zootaxa, 2025),
Circoniscus boitata (MG, Zootaxa, 2025),
Kaatecarcinia apina, K. amazonica, K. karaja (PA, EJT, 2026),
Novamundoniscus anophthalmus, Trichorhina dimorphica, and Pectenoniscus bilobulatus (MS/BA/BA, Annales Zoologici, 2025),
and Iansaoniscus aykuara (MG, Zootaxa, 2025).
ARTHROPODA/MALACOSTRACA/DECAPODA (3)
Aegla charon (SP, Nauplius, 2017), Aegla clotho, A. atropos (SP, nvertebrate Systematics, 2024).
ARTHROPODA/COLLEMBOLA (25)
Pararrhopalites sideroicus (MG, Florida Entomologist, 2014),
Arrhopalites glabrofasciatus, Pseudosinella guanhaensis (MG, Zootaxa, 2018),
Pararrhopalites ubiquum (MG, Neotrop. Entomol., 2018),
10 in Trogolaphysa, 9 in Pseudosinella (PA, MG, SP, Insects, 2020, and PRE-PRINT, 2021),
Troglobentosminthurus luridus (BA, Insects, 2022),
and
Cyphoderus caatinguensis and Pararrhopalites iataganii (RN, Zootaxa, 2024).
ARTHROPODA/INSECTA/ORTHOPTERA (5)
Endecous troglobia (MG, Zootaxa, 2020),
Erebonyx catacumbae (BA, Zootaxa, 2021),
Endecous infernalis (BA, Zootaxa, 2023),
Endecous vitreus (MS, Zootaxa, 2023),
and
Endecous spelaeus (BA/Feira da Mata, Zootaxa, 2025).
ARTHROPODA/INSECTA/COLEOPTERA (11)
Ardistomis ferreirai (PA, Zootaxa, 2018),
Oxarthrius aurora, O. inexpectatus (TO, BA, Zootaxa, 2018),
Metopioxys carajas (PA, Zootaxa, 2019),
Coarazuphium lundi (MG, Zootaxa, 2020),
C. auleri (MT, Studies on Neotropical Fauna and Environment, 2021),
C. bambui (BA, Zootaxa, 2022),
C. xingu, C. xikrin, C. kayapo (PA, Subterranean Biology, 2022),
and
Perigona spelunca (MG, EJT, 2022).
ARTHROPODA/INSECTA/DERMAPTERA (1)
Mesodiplatys falcifer (BA, ZooKeys, 2018).
ARTHROPODA/INSECTA/DIPTERA (1)
Deanemyia maruaga (AM, Mem. Inst. Oswaldo Cruz, 2011).
INSECTA/INSECTA/HEMIPTERA (7)
Spelaeometra gruta, Cephalometra pallida (BA, Tijdschrift voor Entomologie, 2018),
Ferricixius michaeli, F. goliathi (MG, Zootaxa, 2023),
Spelaeometra hypogea (BA, Animals, 2023),
Spelaeodelphax nexus (BA, Subterranean Biology, 2026),
and Pintalia magnaepiprocti (MG, Zootaxa, 2025).
CRANIATA/ACTINOPTERI (1)
Phreatobius sanguijuela (RO/Bolivia, Fauna Cavernicola do Brasil, 2022).
UNDESCRIBED
517 troglobic species remains undescribed, species identifiable as troglobic but not yet formally described, but with certainty or very high signs of troglomorphisms, distributed along following state/references below.
BY REFERENCE AND STATE
Codes in bold, as [MG/4], will be used later to cite these works throughout the group discussion.
GOIÁS
[GO/1] — 12 caves in São Domingos and Posse municipalities — 10 undescribed — Bichuette, ME et al (Subterranean Biology, 2019).
Pseudoscorpiona/Spaleochernes (1), Diplopoda/Pseudonanolene (2), Polydesmida (1), Isopoda/Styloniscidae (1), Isopoda/Dubioniscidae (1), Collembola/Cyphoderus (1), Collembola/Trogolaphysa (2), and Coleoptera/Pselaphinae (1).
[BR/2] — Posse and São Domingos municipalities — 3 undescribed — Bichuette & Gallão (Neotropical Ichthyology, 2021).
Actinopteri/Ituglanis (2), Actinopteri/Phenocorhamdia (1).
MATO GROSSO DO SUL
[MS/1] — 84 caves Bodoquena region and Pantanal region — 30 undescribed — Cordeiro et al. (Biota Neotropica, 2014).
Giradia (1), Oligochaeta (2), Gastropoda/Spiripockia (1), Gastropoda/Pomatiopsidae (1), Onychophora/Peripatopsidae (1), Acari/Hydrozetes (1), Opiliones/Eusarcus (6), Araneae/Ctenidae (1), Paradoxosomatidae (2), Diplopoda/Kantanodesmus (1), Diplopoda/Cryptodesmus (1), Amphipoda/Megagidiella (1), Bathynellacea (1), Isopoda/Oniscidae (1), Collembola/Trogolaphysa (1), Collembola/Entomobryiomorpha (2), Collembola/Cyphoderidae (1), Collembola/Paronellidae (1), Coleoptera/Pachyteles (1), Hemiptera/Dipsocariidae (1), Actinopterii/Rhamdia (1), Actinopterii/Ancistrus (1), and Actinopterii/Trichomycterus (1).
Of the 31 species in the original study, one is here tentatively regarded as described, despite the extremely small possibility that it may not be: Girardia multiverticulata (former Dugesiidae cf. Girardia, ZooKeys, 2015).
[BR/1] — Abismo cave in Jardim municipality — 1 undescribed — Zepon, T et al. (Paíes Avulsos de Zoologia, 2025).
Coleoptera/Hydroscaphidae (1).
PARÁ
[PA/1] — Paraíso cave, Aveiro — 8 undescribed — Souza-Silva, M et al. (Journal of Insect Biodiversity, 2025).
Mollusca/Gastropoda (1), Glomeridesmus (1), Pyrgodesmidae (1), Aphelidesmidae (1), Cryptodesmidae (1), Polydesmida (1), Oniscodesmidae (1), and Escadabiidae (1).
[PA/2] — Carajas region of SE Pará — 122 undescribed — a compilated list of many studies of 473 caves in Canaã dos Carajás (85 caves), Parauapebas (296 caves) and Curionópolis (90 caves) in Trevelin et al. (Ecological Indicators, 2019).
Isotomiella numulifer Deharveng & Oliveira, 1990 (Collembola, Isotomidae), which had been cited in the study for Curionópolis, is here excluded, as there is no evidence that it is a troglobic species (occur in forests, SEE). The study cites Charinus carajas Giupponi & Miranda, 2016 and C. orientalis Giupponi & Miranda, 2016 as troglobic, but this status is rejected for both by [6]. Trogolaphysa dandarae Brito & Zeppelini and T. gisbertae Brito & Zeppelini (SEE) may have been described from populations of Paronellidae sp. 4, although there is no concrete reference indicating whether the populations from Canaã dos Carajás were later described under a different name. Although Amazoniscus spica Campos-Filho, Aguiar & Taiti (SEE), Circoniscus mendesi López-Orozco, Campos-Filho & Bichuette, and C. xikrin López-Orozco, Campos-Filho & Carpio-Díaz (SEE), all Scleropactidae, were described from Pará after that study, there is no evidence that they correspond to Crinocheta sp. 1 or Amazoniscus sp. 1; therefore, all of them are treated here as distinct species. Metopioxys carajas Asenjo, described after the study (SEE), does not, based on the available evidence, correspond to any of the 13 undescribed Staphylinidae species listed in the work (neither the suspect Staphylinidae sp. 8). Coarazuphium kayapo Pellegrini, Ferreira & Vieira and C. xikrin Pellegrini, Ferreira & Vieira, described after the study (SEE), show no evidence of corresponding to any of the 9 undescribed Carabidae species recorded in the survey. Although Paracymbiomma bocaina Rodrigues, Cizauskas & Rheims, 2018 and P. caecus Rodrigues, Cizauskas & Rheims, 2018 could represent Lygromma sp. 2 due to the occurrence of all three in two caves of Canaã dos Carajás (SEE), the evidence goes no further, and therefore we treat the three as distinct species. The wide distribution of Speocera piquira Brescovit, Zampaulo, Cizauskas & Pedroso, 2022 and S. babau Brescovit, Zampaulo, Cizauskas & Pedroso, 2022 provides evidence that neither of them corresponds to any of the four undescribed Ochyroceratidae cited in the study. S. pinima Brescovit, Zampaulo, Cizauskas & Pedroso, 2022 may correspond to Ochyroceratidae sp. or Ochyroceratidae sp. 5, but stronger evidence is lacking (SEE). Very little can be said about Ochyrocera ritxoco Brescovit, Zampaulo & Cizauskas, 2021, O. ritxoo Brescovit, Zampaulo & Cizauskas, 2021 (SEE), O. ungoliath Brescovit, Cizauskas & Mota, 2018 (SEE), and Araneae sp. G . All 11 species are treated here as distinct.
Thus, of the 133 species cited in the study (noting that not all troglobic species from the three municipalities were included in the survey), 3 are excluded, 8 have already been formally described, and 122 are here considered as undescribed troglobic species.
122 original undescribed, 121 remains undescribed:: Oligochaeta (3), Tricladida (1), Geoplanidae (1), Systrophiidae (3), Arachnida (27), Chilopoda/Cryptops (1), Diplopoda (18), Isopoda (11), Collembola (16), Coleoptera (38), Formicidae/Rogeria (1), Hemiptera/Dipsocoridae (1).
Of the 122 species in the original study, one have already been formally described: Clabozoidea sp. 1, as 1 or 2 Kaaetecarcinia (Cardoso, GM et al, EJT, 2025).
TOCANTINS
[TO/1] — 4 caves in Dianópolis — 6 undescribed — Ferreira, RL et al. (CECAV, 2016).
Araneae/Nesticidae (1), Araneae/Symphytogmatidae/Anapistula (1) Polydesmida (1), Styloniscidae (1), Coleoptera/Scydmaeninae (1), Formicidae/Amblyopone (1).
RIO GRANDE DO NORTE
[RN/1] - all caves of Rio Grande do Norte state — 53 undescribed, in a strong conservative list — Bento & Cruz (CaverRNas: o Carste Potiguar, 2025 | VIDEO YOUTUBE GRUTA TRÊS LAGOS).
Hausera (3), Platyhelminthes/Tricladida (1), Oligochaeta (1), Araneae (1), Araneae/Ochyroceratidae (1), Araneae/Oonopidae (1), Araneae/Metagonia (1), Araneae/Prodidomidae (1), Pseudoscorpiones (1), Pseudoscorpiones/Chthoniidae (1), Opiliones (1), Chilopoda/Lithobiomorpha (1), Chilopoda/Dinocryptops (1), Chilopoda/Geophilomorpha (1), Diplopoda/Polydesmida (1), Diplopoda/Pseudonannolene (1), Diplopoda/Cryptodesmidae (1), Diplopoda/ Polyxenida (1), Ostracoda (1), Copepoda (1), Amphipoda/Potiberaba (4), Amphipoda/Seborgia (2), Isopoda/Cirolanidae (5), Isopoda/Trichorhina (1), Isopoda/Styloniscidae (1), Collembola/Arrhopalites (1), Collembola/Cyphoderus (1), Collembola/Pseudosinella (1), Diplura (1), Diplura/Japygidae (1), Insecta/Zygentoma (1), Insecta/Kinnarapotiguara (8), Coleoptera/Carabidae (1) and Coleoptera/Staphylinidae (1).
BAHIA
[BA/1] — 4 caves in Água Clara cave system, Carinhanha municipality — 18 undescribed — Souza-Silva, M et al. (Biodiversity and Conservation, 2021).
Gastropoda/Pulmonata (1), Trombidiformes/Rhagidiidae (1), Araneae/Caponiidae (1), Araneae/Ochyroceceratidae (1), Eukoenenia (1), Diplopoda/Trichopolydesmidae (1), Diplopoda/Pyrgodesmidae (1), Chilopoda/Geophilomorpha (1), Isopoda/Styloniscidae (3), Isopoda/Trichorhina (1), Collembola/Sminthuridae (1), Collembola/Entomobryomorpha (2), Collembola/Symphypleona (1), Insecta/Blattodea(1), Insecta/Nylanderia (1).
[BA/2] — 18 caves from Iuiu and Malhada — 32 undescribed — Cardoso, RC et al. (Biodiversity and Conservation, 2021).
Gastropoda/Scolodontidae (1), Gastropoda (3), Araneae/Ochyroceratidae (1), Pseudoscorpiona/Spelaeobochica (1), Pseudoscorpiona/Chthoniidae (1), Eukoenenia(2), Opiliona/Acantholibinia (1), Opiliona/Zalmoxidae (1), Diplopoda/Pyrgodesmidae (3), Diplopoda (1), Diplopoda/Chelodesmidae (1), Diplopoda/Paradoxosomatidae (1), Diplopoda/Pseudonannolene (1), Chilopoda/Geophilomorpha (1), Chilopoda/Lithobiomorpha (1), Chilopoda/Cryptops (1), Isopoda/Styloniscidae (5), Isopoda/Pectenoniscus (1), Collembola/Arrhopalites (1), Diplura/Projapygidae (1), Insecta/Nicoletiidae (1), Insecta/Blattodea (1), and Coleoptera/Clivinina (1).
[BA/3] — five caves from Igatu region in Andaraí and Lençóis municipalities — 29 undescribed — Gallão, JE et al. (Diversity, 2023).
Gastropoda/Happia (1), Mesostigmata/ Pachylaepidae (1), Mesostigmata/Dithinozeronidae (1), Sarcoptiformes/Oeserchestidae (1), Araneae/Prodidominae (1), Araneae/Ochyrocera (1), Araneae/Metagonia (1), Araneae/Telemidae (1), Pseudoscorpiona/Spelaeochernes (1), Pseudoscorpiona/Pseudochthonius (1), Pseudoscorpiona/Syarinidae (1), Eukoenenia (1), Opiliona/Tricommatidae (1), Diplopoda/Chelodesmidae (1), Diplopoda/Crypturodesmus (1), Chilopoda/Sphendononema (1), Chilopoda/Cryptops (1), Isopoda/Philosciidae (1), Isopoda/Platyartridae (2), Collembola/Verhoeffiella (1), Collembola/Heteromurini (1), Collembola/Troglopedetes (1), Diplura/Projapygidae (1), Insecta/Nicoletiidae (1), Insecta/Blattelidae (1), Coleoptera/Scydmaenidae (1), Coleoptera/Pselaphinae (1), and Actinopteri/Copionodon (1).
.
[BA/4] — Toca do Gonçalo cave in Campo Formoso — 13 undescribed — Sousa Silva & Ferreira (Subterranean Biology, 2016).
15 original undescribed, 14 remains undescribed: Gastropoda/Rotadiscus (1), Araneae/Lygromma (1), Diplopoda/Polyxenida (1), Diplopoda/Oniscodesmidae (1), Chilopoda/Geophilomorpha (1), Isopoda/Scleropactidae (1), Isopoda/Trichorhina (1), Collembola/Arrhopalites (1), Insecta/Ortheziidae (1), Insecta/Nicoletiidae (1), Coleoptera/Clivinina (1), Coleoptera/Dytiscidae (1), and Actinopteri/Rhamdiopsis (1).
Originally, there were 15 undescribed species, two was described latter: Pseudochthonius gonssaloensis (here we consider that this species is the one originally treated as Cthoniidae [sic] sp., Guimarães, L et al., Zoological Studies, 2025), and Erebonix catacumbae (former Phalangospsidae sp. n., Melo & Ferreira, Zootaxa, 2021).
[BA/5] — Carinhanha region — 9 undescribed — Ferreira & Souza-Silva (Diversity, 2023).
10 original undescribed, 9 remains undescribed: Araneae/Palpimanidae (1), Araneae/Ochiroceratidae (1), Araneae/Matta (1), Diplopoda/Siphonophoridae (1), Diplopoda/Trichopolydesmidae (1), Diplura/Projapygidae (1), Coleoptera/Clivinina (1), Coleoptera/Trechinae (1), and Hemiptera/Spelaeometra (1).
Originally, there were 10 undescribed species, but one, Delphacidae sp.1, was described as Spelaeodelphax nexus (Santos, JCCV et al., Subterranean Biology, 2026).
[BA/6] — Pedro Cassiano cave in Carinhanha municipality — 13 undescribed — Souza-Silva, M et al. (Acta Oecologica, 2025).
Araneae/Ochyroceratidae (1), Araneae/Oonopidae (1), Eukoenenia (1), Polydesmida (1), Isopoda/Pectenoniscus (1), Isopoda/Xangoniscus (1), Collembola/Symphypleona (1), Collembola/Neelipleona (1), Collembola/Trogolaphysa (2), Insecta/Blattodea (1), and Coleoptera/Clivinina (2)
[BA/7] — Padre Cave in Santana municipality — 20 undescribed — Pereira Junta, VG et al. (Community, 2025 | VIDEO YOUTUBE).
Nemertea (1), Oligochaeta (1), Gastropoda/Spiripockia (1), Araneae/Ochyroceratidae (1), Opiliona/Escadabiidae (1), Opiliona/Eusacus (1), Eukoenenia (2), Symphyla (1), Isopoda/Platyartridae (1), Isopoda/Styloniscidae (1), Isopoda/Xangoniscus (1), Collembola/Paronellidae (1), Collembola/Poduromorpha (1), Collembola/Arrhopalitidae (1), Insecta/Blattodea (1), Insecta/Coleoptera (1), Insecta/Orthoptera (1), and Actinopteri/Pimelodella (1).
[BR/2] — Coribe, Iuiu, Feira da Mata, Mirangaba and Umburanas — 6 undescribed — Bichuette & Gallão (Neotropical Ichthyology, 2021).
Trichomycterus (2), Rhamdiopsis (4).
ESPÍRITO SANTO
[ES/1] — 15 caves in Santa Teresa, Ecoporanga, Pedro Calvário, Jaciguá, Vargem Alta and Conceição do Castelo municipalities — 10 undescribed — Souza Silva & Ferreira (Subterranean Biology, 2015).
Araneae/Ochyroceratidae (1), Escadabiidae (2), Pseudonannolene (1), Cyrtodesmidae (1), Trichopolydesmidae (1), Isopoda/Trachelypodidae (1), Isopoda/Trichorhina (1), and Zygentoma (2).
MINAS GERAIS
[MG/1] — 15 limestone caves located in the municipalities of Cordisburgo (13 caves) and Curvelo (2 caves) — 35 undescribed — Souza, MVR et al. (International Journal of Speleology, 2021 | SUPPLEMENTARY MATERIAL).
Nemertea (1), Annelida/Hirudinea (1), Mesostigmata (1), Araneae/Ochyroceratidae (1), Araneae/Oonopidae (1), Araneae/Prodidomidae (1), Araneae/Oonopidae (1), Pseudoscorpiona/Chthoniidae (2), Pseudoscorpiona/Pseudochthonius (1), Opiliones/Spinopilar (1), Opiliones/Spaeleoleptes (1), Eukoenenia (1), Diplopoda/Oniscodesmidae (1), Diplopoda/Hypogexenidae (1), Diplopoda/Pyrgodesmidae (1), Diplopoda/Trichopolydesmida (1), Diplopoda/Polydesmida (2), Pauropoda (1), Isopoda/Styloniscidae (2), Isopoda/Pectenoniscus (3), Isopoda/Trichorhina (2), Collembola/Arrhopalites (2), Collembola/Cyphoderidae (1), Diplura/Japygidae (1), Insecta/Zygentoma (1), Coleoptera/Trechinae (1), Coleoptera/Pselaphinae (1), and Coleoptera/Coarazuphuim (1).
[MG/2] — 20 caves from Ibitipoca Estadual Park in Lima Duarte and Santa Rita do Ibitipoca — 5 undescribed — Souza Silva, Iniesta and Ferreira (Subterranean Biology, 2020).
Collembola/Hypogastruridae (1), Diplura/Projapygidae (2), Insecta/Blattodea (1), and Coleoptera/Pselaphinae (1).
[MG/3] — 9 caves in Belo Horizonte, Nova Lima, Ibirité e Brumadinho — 9 undescribed — Zampaulo (33rd CBE, 2015).
Araneae (Tsentinops-1, Lygromma-1), Opiliones/Spinopilar (1), Pseudoscorpiones/Pseudochthonius (1), Diplopoda/Pyrgodesmidae (1), Collembola (3, Pararrhopalites-1, Pseudosinella-1, Trogolaphysa-1), and Coleoptera/Pselaphinae (1).
[MG/4] — 6 caves in Sumidouro State Park, Lagoa Santa municipality — 2 undescribed — Iniesta, LFM et al. (Revista Brasileira de Espeleologia, 2012).
Trombidiformes/Labidostomatidae (1) and Isopoda/Trichorhina (1).
[MG/5] — 14 caves from Peruaçu Caves National Park, in Itacarambi and Januaria municipalities — 2 undescribed — Monte & Bichuette (Biota Neotropica, 2020).
Diplopoda/Oniscodesmidae (1) and Collembola (1).
[MG/6] — Éden cave in Pains municipality — 14 undescribed — Ratton Sousa, P et al. (Revista Brasileira de Espeleologia, 2017).
Opiliones/Paratricommatus (1), Pseudoscorpiones/Chthoniidae (1), Diplopoda/Sphaerodesmidae (1), Symphyla (3), Isopoda/Styloniscidae (2), Copepoda/Harpacticoida (1), Collembola/Arrhopalites (1), Insecta/Blattellidae (1), and Coleoptera/Carabidae (3).
[MG/7] — 7 caves in Arinos municipality — 7 undescribed — Henrique Simões, M. et al. (Revista Brasileira de Espeleologia, 2012).
Turbellaria (1), Annelida/Hirudinea(1), Diplopoda/Oniscodesmidae (1), Diplopoda/Polyxenidae (1), Collembola (1), Isopoda/Trichorhina (1), and Isopoda/Styloniscidae (1).
[MG/8] — 7 caves in Presidente Olegário municipality — 3 undescribed — Zepon (Master’s Thesis/UFSCar, 2015).
4 original undescribed, 3 remains undescribed: Polydesmida (1), Coleoptera/Pselaphinae (1), and Coleoptera/Carabidae (1).
Originally, there were 4 undescribed species, but one, Chthoniidae sp. 4, was described as Pseudochthonius olegario (von Schimonsky, DM et al., Arachnology, 2022).
[MG/9] — Matosinhos municipality — 4 undescribed — Cunha Santos, NM (Master’s Thesis/UFRN, 2020).
Collembola/Trogolaphysa (4).
[MG/10] — Lapa Nova cave, Vazante municipality — 5 undescribed — Pellegrini & Ferreira (Subterranean Biology, 2016).
Araneae/Oonopidae (1), Pseudoscorpiones/Chthoniidae (1), Collembola/Arrhopalites (1), Collembola/Acherontides (1), and Isopoda/Styloniscidae (1).
[MG/11] — cave LS_0005, Santa Bárbara municipality — 1 undescribed — Souza et al. (Zoological Journal of the Linnean Society, 2025) and Souza, MFVR (Anais do 37º Congresso Brasileiro de Espeleologia, 2023).
Eukoenenia (1).
[BR/2] — São Rique de Minas and Cordisburgo — 2 undescribed — Bichuette & Gallão (Neotropical Ichthyology, 2021).
Ituglanis (1), Rhamdiopsis (1).
SÃO PAULO
[SP/1] — 33 caves in Alto Ribeira karst in Iporanga, Apium and Guapiara municipalities — 6 undescribed — Bichuette & Trajano (Subterranean Biology, 2018).
Potamolithus (6).
[SP/2] — three caves in Areias Cave System, Iporanga — 12 undescribed — Souza Silva & Ferreira (Subterranean Biology, 2016).
Girardia (1) Nemertea (1), Araneae/Hahniidae (1), Eukoenenia (1), Cryptodesmidae/Cryptodesmus (1), Oniscodesmidae/Crypturodesmus (1), Pyrgodesmidae/Peridontosmella (1), Isopoda/Trichorhina (2), Collembola/Falsomia (1), Collembola/Cyphoderidae (1), Collembola/Paronellidae (1), and Coleoptera/Carabidae (1).
Of the 13 species in the original study, one is here tentatively regarded as described, despite the extremely small possibility that it may not be: Girardia paucipunctata or G. arenicola (former Dugesiidae sp. n., Zootaxa, 2018).
[SP/3] — Riacho Subterrâneo cave in Itu municipality — 12 undescribed — Bichuette, ME et al. (Neotropical Biology and Conservation, 2017).
Chthoniidae (1), Chelodesmidae (5), Polydesmidae (2), Geophilidae (1), Symphyla (1), Copepoda (1), Collembola/Entomobryiomorpha (1).
[SP/4] — two caves in Bulhas d'água regio, Iporanga municipality — 1 undescribed — Zepon, T et al. (Espeleo-Tema, 2019).
Paradoxosomatidae (1).
[BR/2] — Juquiá municipality — 1 undescribed — Bichuette & Gallão (Neotropical Ichthyology, 2021).
Pimelodella (1).
PARANÁ
[PR/1] — 12 limestone caves in Itaiacoca formation in NE Paraná state in Dr. Ulysses, Castro, Cerro Azul and Sengès municipalities — 13 undescribed — Mata Melo, L et al. (37th CBE, 2023).
Gastropoda (1), Araneae/Prodidomidae (1), Opiliones (2), Palpigrada (3), Chthoniidae (1), Crypturodesmus (1) Pyrgodesmidae (3) and Isopoda/Trichorhina (1).
UNVAILABLE ARTICLES
48 caves Bambuí system in Minas Gerais state — unknown number of undescribed — Rabelo, LM et al. ( Biodiversity and Conservation, 2018).
Unknown list.
The Proceedings of the 19th International Congress of Speleology (ICS/vol. VI | ICS/vol. VII, 2025) include several preliminary accounts of forthcoming studies, notably inventories of new species with strong troglobic potential in Brazil. These comprise Chthoniidae gen. nov. 1 (vol. VII, p129) from the Carajás Complex, Pará; four potential new species of Isopoda/Cirocniscus (vol. VII, p119), also from the Carajás Complex; four new potentially troglobic species of Coleoptera/Noteridae (vol. VII, p35); one species of Orthoptera/Eidmanacris (vol. VI, p95) from Toca da Raposa 2 Cave, Murici, Alagoas; seven species of Orthoptera/Aracambiae (vol. VI, p131); one species of Litoblatta (Blattodea: Blattellidae; vol. VI, p279) from Gruta do Éden Cave, Pains, Minas Gerais; several species of Dytiscidae from the Carajás Complex (vol. VII, p266); notes on the prospection of new species in Santana, Santa Maria da Vitória, and Canápolis, Bahia (potentially 41 troglobic species; vol. VI, p238); one hundred caves in the Dois Irmãos region (MG, 20 potential troglobic species; vol. VII, p134); the global troglobic potential of Diplura (vol. VII, p189); and a list of potential areas for future prospection in Strategic Sites for Cave Prospecting and Non-Troglobic Faunal Surveys in Brazil (vol. VI, p139). Undescribed species mentioned in these inventories are not considered here.
BY GROUP
The highest numbers by undescribed troglobic in Brazil includes Insecta (103), Diplopoda (82), Collembola (63), Isopoda (61), Araneae (44), and Opiliones (29).
PLATYHELMINTHES (7)
RN (4), PA (2), MG (1).
NEMERTEA (3)
BA (1), SP (1), MG (1).
ANNELIDA (10)
PA (3), MS (2), MG (2), RN (1), BA (1).
MOLLUSCA (20)
BA (8), SP (6), PA (3), MS (2), PR (1).
ONYCHOPHORA (1)
MS (1).
ARTHROPODA
ARACHNIDA
Acari (9) ‣ BA (4), MG (2), PA (2), MS (1).
Araneae (46) ‣ BA (14), PA (12), MG (7), RN (5), TO (2), ES (1), SP (1), MS (1), PR (1).
Pseudoscorpionida (21) ‣ MG (6), BA (5), PA (5), RN (2), SP (1), GO (1), PR (1).
Palpigrada (12) ‣ BA (7), PR (3), MG (2), SP (1).
Schizomida (1) ‣ PA (1).
Opiliones (27) ‣ PA (8), MS (6), BA (5), MG (4), ES (2), RN (2), PA (2).
MYRIAPODA
Pauropoda (1) ‣ MG (1).
Symphyla (5) ‣ MG (3), BA (1), SP (1).
Diplopoda (81) ‣ PA (24), BA (16), MG (12), SP (11), MS (4), PR (4), RN (4), ES (3), GO (3), TO (1).
Chilopoda (15) ‣ BA (7), RN (3), SP (1), PA (1).
OLIGOSTRACA
Ostracoda (1) ‣ RN (1).
MALACOSTRACA
Amphipoda (7) ‣ RN (6), MS (1).
Bathynellacea (1) ‣ MS (1).
Isopoda (61) ‣ BA (21), MG (13), PA (11), RN (7), SP (2), ES (2), GO (2), MS (1), TO (1), PR (1).
COPEPODA
Copepoda (5) ‣ RN (1), SP (1), BA (1), MG (1).
HEXAPODA
Collembola (63) ‣ MG (16), BA (16), PA (16), MS (5), SP (4), RN (3), GO (3).
Diplura (9) ‣ MG (3), BA (3), RN (2).
Insecta (103)
Zygentoma ‣ BA (3), ES (2), MG (1), RN (1).
Coleoptera ‣ PA (38), MG (10), BA (10), RN (2), MS (2), SP (1), GO (1), TO (1).
Blattodea ‣ BA (5), MG (2).
Hemiptera ‣ RN (8), BA (2), MS (1), PA (1).
Orthoptera ‣ BA (1).
Hymenoptera ‣ BA (1), TO (1), PA (1).
ACTINOPTERI (18)
BA (9), MS (3), GO (3), MG (2), SP (1).
BRAZILIAN TROGLOBIC BY GROUP
Highest diverse group in troglobic species in Brazil are: Isopoda (140), Collembola (103), Diplopoda (96), Coleoptera (89), Araneae (79), Pseudoscorpiona (53), Opiliones (44), Actinopteri (39), Palpigradi (33), Gastropoda (30), Amphipoda (30), Chilopoda (21), Platyhelminthes (18), Hemiptera (22), Amblypygi (11) and Diplura (9).
BRAZILIAN TROGLOBIC BY STATE
14 troglobic species occur in more one state: Phreatobius sanguijuela (RO, Bolivia),
Phreatobius cisternarum (AP, PA),
Potiicoara brasiliensis (MT, MS),
Peridontodesmella alba (SP, PR),
Potieraba porakuara (RN, CE),
Brasilana spelaea (RN, CE),
Acherontides eleonorae (SP, PR),
Ideoroncus cavicola (SP, PR),
Pseudochtonius strinatii (MG, SP, PR),
P. biseriatus (PE, MG),
Spelaeochernes dubius (MS, MS, SP),
S. eleonorae (MS, GO, MS, SP),
S. gracilipalpus (MG, SP), and
S. popeye (BA, SE).
If you count described and undescribed species state endemic, the numbers are: Bahia (257), Minas Gerais (196), Pará (174), São Paulo (67), Rio Grande do Norte (63), Mato Grosso do Sul (44), Goiás (26), Paraná (17), Espírito Santo (11), Tocantins (9), Mato Grosso (4), Ceará (2), Santa Catarina (2), Rio de Janeiro (2), Piauí (1), Amazonas (1), Rondonia (1) and Sergipe (1). Amapá no has endemic troglobic.
If we count all the species (including non state endemic), we have, the numbers are Bahia (258), Minas Gerais (200), Pará (175), São Paulo (73), Rio Grande do Norte (66), Mato Grosso do Sul (47), Goiás (26), Paraná (21), Espírito Santo (11), Tocantins (9), Ceará (5), Mato Grosso (5), Santa Catarina (2), Rondonia (2), Rio de Janeiro (2), Amazonas (1), Piauí (1), Sergipe (2) and Amapá (1).
Undescribed for state: Bahia (141), Pará (129), Minas Gerais (89), Rio Grande do Norte (53), São Paulo (32), Mato Grosso do Sul (31), Paraná (13), Goiás (13), Espírito Santo (10), Tocantins (6). 518 spp., all state endemic.
TROGLOBIC BY MUNICIPALITY
Most distributions followed the original descriptions of the species, or local inventories. Another important source consulted was Pires et al. (CBE, 2015) and [1]. 162 municipalities has troglobic in Brazil based on our survey. The species that was identified in the most municipalities was Trogolaphysa bellinii (Collembola/Paronellidae), having been identified in 10 municipalities in Minas Gerais state.
AM [1] — Presidente Figueredo (1).
RO [2] — Porto Velho (1) and São Francisco do Guaporé (1).
AP [2] — Amapá (1) and Macapá (1).
PA [13] — Parauapebas (89), Curionópolis (52), Canaã dos Carajás (41), Aveiro (10), São Felix do Xingu (3), Altamira (2), Medicilândia (3), Ananindeua (1), Belém (1), Benfica (1), Brasil Novo (1), Eldorado dos Carajás (1), and Salvaterra (1).
PI [1] — Coronel José Dias (1).
CE [3] — Quixerê (2), Santa Quitéria (1), and Ubajara (1).
RN [5] — Felipe Guerra (37), Dix-Sept Rosado (13), Baraúna (8), Apodi (4), and Jandaira (1).
SE [3] — Japaratuba (1), Lagarto (1), and Laranjeiras (1).
BA [29] — Carinhanha (62), Andaraí (38), Iuiu (34), Campo Formoso (29), Santana (28), Malhada (20), Coribe (12), Feira da Mata (7), Iraquara (7), Itaetê (6), Serra do Ramalho (6), Morro do Chapéu (4), São Desidério (4), Palmeiras (3), Paripiranga (3), Varzea Nova (3), Ituaçu (2), Lençóis (2), Miranguara (2), Santa Maria da Vitória (2), Bom Jesus da Lapa (1), Canálopis (1), Curaçá (1), Maria da Vitória (1), Mucugê (1), Nova Redenção (1), Ourolândia (1), Pau Brasil (1), Umburana (1).
TO [4] — Dianópolis (6), Aurora de Tocantins (1), Lagoa da Confusão (1), and Natividade (1).
MT [4] — Nobres (2), Curvelândia (1), Rosario do Oeste (1), and Diamantino (1).
GO [8] — São Domingos (18), Posse (5), Mambaí (3), Buritinópolis (1), Corumbá de Goiás (1), Guarani de Goiás (1), Nova Roma (1), Pedro Bernardo (1).
MG [54] — Cordisburgo (42), Itacarambi (17), Pains (17), Matozinhos (11), Rio Acima (11), Belo Horizonte (10), Mariana (10), Itabirito (9), Arinos (7), Brumadinho (7), Pedro Leopoldo (7), Barão de Cocais (6), Morro do Pilar (6), Nova Lima (6), Presidente Olegário (6), Sete Lagoas (6), Vazante (6), Conceição do Mato Dentro (5), Lima Duarte (5), Montes Claros (5), Santa Bárbara (5), Caete (4), Luislandia (4), Prudente de Morais (4), Curvelo (3), Lagoa Santa (3), Ibiracatu (2), Itambé do Mato Dentro (2), Monjolos (2), Prados (2), Rio Pardo de Minas (2), São Gonçalo do Rio Abaixo (2), São Roque de Minas (2), Arcos (1), Capim Branco (1), Carai (1), Catas Altas (1), Dores de Guanhaes (1), Jaiba (1), Jequitaí (1), Juvenilia (1), Moeda (1), Montalvania (1), Ouro Preto (1), Piumhi (1), Presidente Juscelino (1), Santa Maria de Itabira (1), São João da Lagoa (1), São João da Lapa (1), São João da Ponte (1), São Sebastião do Rio Preto (1), Unaí (1), and Vespasiano (1).
ES [5] — Vargem Alta (5), Ecoporanga (2), Santa Teresa (2), Conceição do Castelo (1), and Pedro Calvário (1).
RJ [2] — Cantagalo (1) and Cambuci (1).
SP [12] — Iporanga (52), Itu (12), Eldorado (3), Guapiara (3), Itirapina (2), Altinopólis (1), Analandia (1), Apiai (1), Ipeuna (1), Juquiá (1), Ribeirão Grande (1), and Ribeira (1).
MS [5] — Bodoquena (24), Bonito (24), Jardim (6), Porto Murtinho (3), and Corumbá (3).
PR [8] — Dr. Ulysses (9), Castro (7), Cerro Azul (4), Sengés (4), Rio Branco do Sul (3), Adrianópolis (2), Almirante Tamandaré (1), and Ponta Grossa (1).
SC [1] — Botuverá (2).
RS [1] — Santa Tereza (1).
RANKING BY MUNICIPALITY
Parauapebas (89), Carinhanha (62), Curionópolis (52), Iporanga (52), Cordisburgo (42), Canaã dos Carajás (41), Andaraí (38), Felipe Guerra (37), Iuiu (34), Campo Formoso (29), Santana (28), Bodoquena (24), Bonito (24), Malhada (20), São Domingos (18), Itacarambi (17), Pains (17), Governador Dix-Sept Rosado (13), Coribe (12), Itu (12) — Matozinhos (11), Rio Acima (11) — Aveiro (10), Belo Horizonte (10), Mariana (10) — Dr. Ulysses (9), Itabirito (9) — Baraúna (8) — Arinos (7), Brumadinho (6), Castro (7), Feira da Mata (7), Iraquara (7), Pedro Leopoldo (7), Serra do Ramalho (7) — Barão de Cocais (6), Dianópolis (6), Itaetê (6), Jardim (6), Morro do Pilar (6), Nova Lima (6), Presidente Olegário (6), Sete Lagoas (6), Vazante (6) — Conceição do Mato Dentro (5), Lima Duarte (5), Montes Claros (5), Posse (5), Santa Bárbara (4), Vargem Alta (5) — Apodi (4), Caete (4), Cerro Azul (4), Luislandia (4), Morro do Chapéu (4), Prudente de Morais (4), São Desidério (4), Sengés (4) — Caete (3), Corumbá (3), Curvelo (3), Eldorado (3), Guapiara (3), Lagoa Santa (3), Mambaí (3), Palmeiras (3), Paripiranga (3), Porto Murtinho (3), Rio Branco do Sul (3), São Felix do Xingu (3), Varzea Nova (3) — Adrianópolis (2), Altamira (2), Botuvera (2), Ecoporanga (2), Ibiracatu (2), Itambé do Mato Dentro (2), Itirapina (2), Ituaçu (2), Lençóis (2), Medicilândia (2), Miranguara (2), Monjolos (2), Mossoró (2), Nobres (2), Prados (2), Quixerê (2), Rio Pardo de Minas (2), Santa Maria da Vitória (2), Santa Teresa (2), São Gonçalo do Rio Abaixo (2), São Roque de Minas (2) — Almirante Tamandaré (1), Altinopólis (1), Amapá (1), Analandia (1), Ananindeua (1), Apiai (1), Arcos (1), Aurora de Tocantins (1), Belém (1), Benfica (1), Bom Jesus da Lapa (1), Brasil Novo (1), Canálopis (1), Cantagalo (1), Cambuci (1), Conceição do Castelo (1), Capim Branco (1), Caraí (1), Catas Altas (1), Coronel José Dias (1), Corumbá de Goiás (1), Curaçá (1), Curvelândia (1), Diamantino (1), Dores de Guanhaes (1), Eldorado dos Carajás (1), Guarani de Goiás (1), Ipeuna (1), Jaiba (1), Jandaira (1), Japaratuba (1), Jequitaí (1), Juquiá (1), Juvenilia (1), Lagarto (1), Lagoa da Confusão (1), Laranjeiras (1), Macapá (1), Maria da Vitória (1), Moeda (1), Montalvania (1), Mucugê (1), Natividade (1), Nova Redenção (1), Nova Roma (1), Ouro Preto (1), Ourolândia (1), Pau Brasil (1), Pedro Bernardo (1), Pedro Canário (1), Piumhi (1), Ponta Grossa (1), Porto Velho (1), Presidente Figueredo (1), Presidente Juscelino (1), Ribeirão Grande (1), Ribeira (1), Rosario do Oeste (1), Salvaterra (1), Santa Quitéria (1), Santa Maria de Itabira (1), Santa Tereza (1), São Francsco do Guaporé (1), São João da Lagoa (1), São João da Lapa (1), São João da Ponte (1), Ubajara (1), Unaí (1), Umburana (1) and Vespasiano (1).
MAP OF TROGLOBIC
Below is the distribution map of troglobic in Brazil, represented by black dots, and phreatic species by red dots. Each dot may correspond to more than one cave and/or municipality.

BRAZILIAN TROGLOBIC (BLACK DOTS) AND PHREATIC (RED DOTS) METAZOA IN POLITICAL SUBDIVISIONS AT THE MUNICIPAL LEVEL (N, WC, MA, PI, CE, BA), MICROREGIONS (RN, PB, PE, MG, SP, PR, RS), and MESOREGIONS (AL, SE, RJ, ES, SC)
CAVE FUNGI IN BRAZIL
Cave fungi represent a diverse array of species that underwent speciation beyond their subterranean confines, providing several benefits to the biosystems they inhabit. 292 fungal genera belonging to six phyla (Ascomycota, Basidiomycota, Basidiobolomycota, Chytridiomycota, Mucoromycota, and Mortierellomycota) have been recorded, and a few operational taxonomic units have been identified as Rozellomycota and Kickxellomycota. Nearly half of the cave fungi known worldwide are found in only 30 caves in Brazil (José Prazeres, Fungal Biology Reviews, 2025). Additional notes on the mycobiota in caves of the Carajás Complex can be found in Silva, A. et al. (ICS 2025, vol. VII, p. 195).
LAND PLANTS FROM CAVES IN BRAZIL
Bryophytes comprise three distinct evolutionary lineages, liverworts (Marchantiophyta), mosses (Bryophyta) and hornworts
(Anthocerotophyta), considered extremophile organisms because they are capable of growing in extremely dry or cold environments or even with low light intensity such as caves, where bryophytes may be the most commonly found plants. Considering Brazilian
caves, there are few records of bryophytes in the literature — a total of 1,145 records of bryophyte occurrences were observed in 21 Brazilian caves, covering 15 states, with a total of 277 species have been identified, comprising 75 species of Marchantiophyta, 200 of Bryophyta, and two of Anthocerotophyta (ICS/vol. VII, pg. 266).
ADDITIONAL STUDIES, SOURCES, AND REFERENCES
Some inventories result in confirmation of the absence of troglobic in some locations, such as Martins (RN, Araujo et al., Espeleo-Tema, 2017). Some works also maybe inconclusive in quantity. Ferreira, Zampaulo & Souza-Silva (Chapter Book, 2022) cites 50 spp. in the Pains karst, only 6 described until the publication of the article, suggesting 44 spp. new undescribed. As the article does not bring a listing by group, here it is not considered to accept these numbers. Simões et al. (Subterranean Biology, 2015) cites 33 troglobic/trogomorphic species in Minas Gerais state, however, without discriminating which are exact troglobic, we decided not to count these taxa.
Some studies address the troglobic fauna of the mesovoid shallow substratum in cangas of Minas Gerais (e.g. Sabach, L, et al, Scientific Reports, 2024), with some interesting records. However, we do not analyze or explore studies in mesovoid environments here.
Of all the phyla that evolved troglobic species, Brazil has representatives of all except Cnidaria, Syndermata and Nematoda. Lineages with troglobic unknown for troglobic in Brazil includes, by phyllum:
Cnidaria (Balkans).
Themnocephalida (S Europe), Prorhyhchidae (South Africa).
Nematoda (Colombia and Europe).
Syndermata/Acanthocephala (USA).
Sabellida (NE Italy, Slovenia, Croatia, Bosnia & Hercegovina) and Phyllodocida (SE Mexico and Caribbean, Canary Islands, Oman, Socotra, W. Australia, Papua New Guinea, New Zealand and Fiji).
Arachnida/Opilioacarida (Cuba, Thailand and Vietnam), Arachnida/Schizomida (Cuba, Jamaica, Belize, Mexico, USA, Ecuador, and Australia), Arachnida/Thelyphonida (Laos), Arachnida/Ricinulei (Venezuela, Belize, Cuba and Mexico).
Branchiopoda (Spain, Romania, France, Slovenia, Bosnia and Herzegovina), Anaspidaceae (Australia and Tasmania), Thermosbaenacea (USA, Cuba, Puerto Rico, Italy, France, Thailand, Cambodia, W Australia), Mysida (several places worldwide), Decapoda/Caridea (widely worldwide), Decapoda/Astacida (Papua New Guinea, Cuba, Mexico, North America), Decapoda/Brachyura (widely worldwide).
Insecta/Psocoptera (Venezuela), Insecta/Lepidoptera (Philippines, Hawaii and Colombia).
Amphibia (USA and Balkans in Europe).






CARDS
This text contains three types of cards: standard cards for each species; yellow cards intended for species to be added; and gray cards intended to close the rows of species.


YELLOW CARDS (SPECIES TO BE ADDED) AND GRAY CARDS (CLOSE ROWS OF SPECIES)
The final standard format of this website is the card, according to the specifications below. Many of the cards are still in older and intermediate formats, and gradually all will be converted to the standard format.

1 PHYLUM PORIFERA ‣ 4 spp. of Porifera was known worldwide as troglobic: Eunapius subterraneus Sket & Velikonja 1984, found in the Ogulin caves, Croatia[2]; Arinosaster patriciae Volkmer-Ribeiro, Tavares-Frigo, Ribeiro & Bichuette, 2021, collected in Sumidouro do Rio Claro sinkhole in the Parecis System, Mato Grosso state, WC Brazil (Neotropical Biology and Conservation, 2021); Racekiela cavernicola Volkmer-Ribeiro, Bichuette & Machado, 2010, collected in Lapa dos Brejões Cave, northern Bahia state, NE Brazil (Volkmer et al., Neotropical Biology and Conservation, 2010); and a yet undescribed species, discovered in San Luis Potosí state, C Mexico (Legendre et al., Subterranean Biology, 2023).


2 PHYLUM CNIDARIA ‣ many records of Cnidaria in freshwater caves can be found in Zagmaster et al. (Speleobiology, 2011), including in Mexico, USA, Australia, and several Central European countries. Velkovrhia enigmatica Matjašić & Sket, 1971 (Bougainvilliidae, Hydrozoa, PHOTO), from the Dinarides in the Balkan Peninsula, is the only known troglobic freshwater Cnidaria. It has been reported from five caves, in Slovenia (3), Croatia (1), and Bosnia & Hercegovina (1), according to Magmajster (Natura Sloveniae, 2003). Unfortunately, it is not listed among freshwater Cnidaria in [3].
3 PHYLUM SYNDERMATA ‣ the current phylum that encompasses the former phyla Rotifera and Acanthocephala. To date, only one species in this group has been reported as troglobic: Dendronucleata americana Moravec & Huffman, 2000 (Eoacanthocephala), a parasite of Typhlomolge rathbuni Stejneger (Amphibia Caudata), known only from Texas, USA[33] (Moravec & Huffman, Folia Parasitologica, 2000).
4 PHYLUM PLATYHELMINTHES ‣ our taxonomic survey identifies 182 species of Platyhelminthes restricted to subterranean environments worldwide, all Rhabditophora/Trepaxonemata, with a peak in taxonomic diversity in the USA, largely due to parasitic species associated with other taxa considered troglobic. In this context, a total of six troglobitic lineages of Platyhelminthes are known.
Four of these lineages have a very limited distribution: Amplimatricata (1, Prorhynchidae, Geocentrophora cavernicola Carpenter, 1970, from Kentucky, Virginia and West Virginia), Euneoophora/Proseriata (1, South Africa, Subterranean Biology, 2020), Euneoophora/Rhabdocolea (13 spp., all in Balkans), Euneoophora/Acentrosomata/Neodermata (Proteocephalus poulsoni Whitaker & Zober, 1978, from Kentucky, in Cestoda; Brachycoelium longleyi Moravec & Huffman, 2000 in Trematoda, from Texas, parasitic in Typhlomolge rathbuni Stejneger, Caudata, Folia Parasitologica, 2000).
The last group, more widely distributed and the only one with species in the Neotropics and Asia, is Euneoophora/Acentrosomata/Tricladida, with 3 in Africa[7], 99 in Europe[2][21], 4 in Russia[21], SE Asia[22], 31 in North America[33], 5 in Mexico[5], 2 in Guatemala[31], 1 in Jamaica[24] and 18 in Brazil.

PHYLOGENY OF TROGLOBIC PLATYHELMINTHES
SOME NOTES
Tricladida in the USA includes (3:4/)31 spp. in Dendrocoelidae (two in Dendrocoelopsis), Kenkiidae (21 in Kenkia and Sphalloplana), and Planariidae (8 in Phagocata). A record of a Temnocephalidan species associated with a crab in Papua New Guinea, previously considered troglobic[22], is here rejected. No troglobic Platyhelminthes have been reported from China[2].
BRAZIL
All known Brazilian troglobic Platyhelminthes belong to the order Tricladida, spanning all three of its clades: Continenticola (10, Girardia-Dugesiidae and one Geoplanidae), Maricola (1), and Cavernicola (4, VIDEO), and three undeterminated. 7 undescribed species have also been cited, 2 from Pará [PA/2], 4 in Rio Grande do Norte [RN/1] and 1 in Minas Gerais [MG/7].
Brazil is the only country with troglobic representatives in all three Tricladida clades. Sluysia triapertura Leal-Zanchet & Souza, 2018 represents the first Maricola triclad recorded from a freshwater cave habitat (Souza et al., Invertebrates Systematics, 2018).
UNDETERMINED ORDER/FAMILY




CAVERNICOLA




CONTINENTICOLA/DUGESIIDAE










CONTINENTICOLA/GEOPLANIDAE


MARICOLA


5 PHYLUM ANELLIDA ‣ based on our surveys, three clades within Annelida has troglobic: Sabellida, Phyllodocida and Clitellata. Some families have adapted to living in the water column of marine/anchialine caves, including the normally benthic Protodrilidae, Nerillidae, and Scalibregmatidae. The aptly named genus Troglochaetus (Nerillidae) is also found in the hyporheic zone of caves, wells and springs. Troglochaetus has rudimentary parapodial cirri and, like all nerillids, development is direct, either via anexternal brood or in cocoons (Glasby et al., Diversity, 2021). Numerous marine species live in conditions similar to cave conditions: inside the bottom material, under stones and in the rock fissures. Morphological adaptations to cave environment such as the absence of body pigmentation and lack of eyes are rarely observed in polychaetes; principally therefore they can be classified as stygobionts only on the basis of their habitat [2].

PHYLOGENY OF TROGLOBIC ANNELIDA
Our survey point to 172 known troglobic Annelida, being 156 Clitellata, 1 Sabellida, and 15 in Nemanereis.
SABELLIDA
Sabellida has one full troglobic, Marifugia cavatica Absolon & Hrabe, 1930 (Serpulidae), a eyeless species known from caves situated in Dinaric Karst in NW Italy, Slovenia, Croatia, and Bosnia & Hercegovina. Even though some localities are situated few kilometers from the seashore this species has never been found in brackish or salt water[2].
PHYLLODOCIDA
Phyllodocida includes 15 troglobic species, all in Namanereis (however, sometimes inconcluse be troglobic, C.J.Glasby et al., ZJLS, 2014), in Mexico (N. cavernicola Solís-Weiss & Espinasa, 1991 — some populations extreme continental, found in cave pools above 1600 m asl and over 170 km from the coastline in Guerrero state, Subterranean Biology, 2017), Caribbean (N. hummelincki, N. minuta, N. serratis, N. stocki, N. sublittoralis, N. christopheri, Subterranean Biology, 2017), Canary Islands (N. llanetensis and N. canariarum, Subterranean Biology, 2020), Oman (N. araps, Beaufortia, 1997), Socotra (N. socotrensis, N. gease, ZJLS, 2014), W Australia (N. pilbarensis, ZJLS, 201), Papua New Guinea (N. berone, RAMS, 1999), New Zealand and Fiji (N. tiriteae, dubious troglobic status, RAMS, 1999).
CLITELLATA
Aquatic members have a habitus that makes them pre-adapted to live in the subterranean environment. In addition, they do not exhibit any peculiar morphological adaptations to subterranean life that can be seen in other subterranean organisms (loss of eyes, elongation of appendages and body, loss of pigmentation, increase in sensory structure). Therefore, stygobitic nature in Clitellata can only be inferred from their exclusive presence in the subterranean environment. Despite their morphological pre-adaptation, few aquatic Clitellata are exclusively present in groundwater.
Our survey listed 156 troglobic species of Clitellata worldwide (with a single overlopping species in Europe no URSS and Georgia), 84 in Europe non former URSS (Tubificidae-14, Enchytreidae-10, Haplotaxidae-5, Parvidrilidae-1, Lumbriculidae-27), 6 in Africa (Guinea-3, Algeria-2, Morocco-1), 10 in former URSS, 2 in Oman, 1 in Iran (Erpobdella borisi), 3 in China, 2 in Japan, 1 in Australia, 2 in New Zealand, 22 in USA/Canada, 4 in Mexico (2 Eodrilus, 2 Branchiobdellida), 3 in Caribbean, 5 in Cuba, 1 in Venezuela, 2 in Ecuador and 9 in Brazil.
After [20], was added 2 spp. in Haplotaxidae from USA, 7 in Lumbriculidae, 4 from Africa, 2 from Mexico, and 9 from Brazil.
INDETERMINATED FAMILY
Includes all 7 undescribed species from Brazil, in Pará [PA/2], Rio Grande do Norte [RN/1], Bahia [BA/7] and Mato Grosso do Sul [MS/1] states.








PARVIDRILLIDA/PARVIDRILLA
Two troglobic spp. in Parvidrilus, P. spelaeus Martinez-Ansemil (2002) in Italy and Slovenia, and P. strayeri Erseus, 1999 in Alabama, USA[33] (Proceedings of the Biological Society of Washington, 1999).
ENCHYTRAEIDA/ENCHYTRAEIDAE
13 troglobic species worldwide, 10 in Europe (Poland, Hungary, Romania, Greece), two in Ecuador: Marionina ecuadoriensis Righi (1981) and Hemienchytraeus mauriliae Righi (1981), and Metataxis carnivorus Omodeo, 1987 from Guinea[7].
TUBIFICIDA/NAIDIDAE
Former Tubificidae. (19/)53 troglobic spp., being nine spp. in New World: Phallodriloides macmasterae Erséus, 1986 in Bermuda; four Pristina and Clitellio cavernicolus Botea, 1983 in Cuba; Dero haitiensis Dumnicka, 1986 and Spirospermoides stocki Dumnicka 1983 in Haiti and adjacent islands; and Krenedrilus papillatus Dumnicka, 1983 in Venezuela; also 38 in Europe (Portugal, Spain, France, Germany, Switzerland, Italy, Poland, Czech Republic, Slovenia, Macedonia, Greece and Turkyie); 5 more species, from Russia, Uzbekisthan, Abkhazia and Georgia[21]; and Pristina jenkinae (Stephenson, 1931) from Algeria[7]. Gianius aquaedulcis Hrabě, 1960 is rejected in USA/New York, and accepted here only for Europe (Polish Journal of Ecology, 2009).
TUBIFICIDA/PHREODRILIDAE
(3/)6 troglobic spp.[20], in New Zealand (2, including Phreodrilus beddardi, A Guide to the Groundwater Invertebrates of New Zealand, 2003), Oman (2), Morocco (1) and Australia (1).
HAPLOTAXIDA/HAPLOTAXIDAE
(3/)9 troglobic spp., known in Europe (5, Spain, France and Bulgary)[20], Japan (1)[20], Guinea (2)[20], and USA (Haplotaxis brinkhursti Cook, 1975, in West Virginia)[25][33].
CRASSICLITELLATA/ACANTHODRILLIDAE
Two troglobic known, both Eodrilus from Mexico[14].
CRASSICLITELLATA/LUMBRICIDAE
(2/)2 spp. (Eophila hypogea Malevics, 1947 and Archaeodrilus cavaticus Czerniavsky, 1880), cited for Georgia and Abkhazia[21], and one known from Algeria (Trichodrilus allobrogum Claparède, 1862)[7].
LUMBRICULIDA/DORYDRILLIDAE
Two troglobic spp., in France and Germany[20].
LUMBRICULIDA/LUMBRICULIDAE
(8/)43 troglobic spp., in West Europe (28, UK, Spain, France, Belgium, Germany, Switzerland, Italy, Estonia, Slovenia, Czech Republic, Croatia, Serbia, Bosnia-Herzegovina, Macedonia, Romania)[20], Abkhazia (undescribed species, counted here as only one)[21], and Japan (1), plus (5/)14 troglobic spp. from USA in Virginia, Alabama, Tennessee in E, Oregon and California[33].
HIRUDINA
At least 10 spp. of troglobic leeches are listed, in three families, being Haemopis caeca Klemm & Sarbu, 1998 (Haemopidae) from Dobrogea in Romania, Erpobdella absoloni Johansson, 1913 (sometimes as Dina absoloni) in N Italy–Balkans–Turkey–Georgia[21], E. krasensis Sket, 1968, and E. mestrovi Kerovec, Kučinić & Jalžić, 1999 from Croatia (Cichoka, 2015), E. underminated spp. from Abkhazia[21], E. borisi Cichocka & Bielecki from NW Iran and Iraq (Hallaq, ZANCO Journal of Pure and Applied Sciences, 2023), two in China ([2] | Novataxa), and two in Brazil, both undescribed collected in C Minas Gerais state [MG/1] and [MG/7] - if confirmed, they will be the firsts troglobic Hirudinea described for the New World.
Haemopidae, Haemadipsidae and Erpobdellidae, usually exhibiting two specific morphological features, namely milky-white coloration and lack of eyes or eye pigment (Cichocka et al., Zootaxa, 2015). Possibly undescribed species, probably Erpobdellidae, occur in caves of China and the USA (Sket & Trontelj, Hydrobiologia, 2006), non counted here. Leiobdella jawarerensis Richardson, 1974 (Haemadipsidae) possess five pairs of eyes, which is the only pigmented part on its body, inhabiting the aphotic zone of a caves in Papua New Guinea (Cichoka, 2015), however is no accepted here as troglobic.


BRANCHIOBDELLIDA
Stygobitic, usually ectoparasites of crustaceans, some of them (similarly to flatworms Themnocephalida) were found on one to three host species[2]. 8 troglobic known, two in Mexico (Mexican Cave Fauna, 2022) and six in USA[33], all Cambarincola in Alabama, Florida, Indiana, Kentucky and Tennessee.
6 PHYLUM NEMERTEA ‣ two out of 20 known freshwater Nemertea species are possibly troglobic (stygobiotic)[2], both accepted hereas troglobic: Prostoma puteale Beauchamp, 1932, found in France, Switzerland, and Germany, and P. hercegovinense Tarman, 1961, found in Bosnia & Herzegovina. Additionally, one species has also been described from New Zealand, and three possibly troglobic Nemertea are known in Brazil: one from the Alto do Ribeira region in São Paulo state [SP/2], one from central Minas Gerais state [MG/1], and one from Padre Cave in Bahia state [BA/7]. If confirmed, these would be the first troglobic Nemertea described from the New World.




7A MOLLUSCA/BIVALVIA ‣ bivalvia includes six troglobic species known worldwide: four Euglesa (Euglesidae) from Eastern Europe[21] — in Russia (1), Abkhazia (2), and Georgia/Armenia (1); Congeria kusceri Bole, 1962 (Dreissenidae, Total Croatia News), known only from Slovenia to SW Bosnia and S Dalmatia; and Eupera troglobia Simone & R.L.Ferreira, 2022 (Sphaeriidae), restricted to western Tocantins state in central Brazil, representing the only known troglobic Bivalvia in the Western Hemisphere.


7B MOLLUSCA/GASTROPODA ‣ all terrestrial troglobic snails are all Heterobranchia/Pulmonata, except for the Caenogastropoda Pholeoteras euthrix Sturany, 1904, known from caves in S Croatia, southern Herzegovina and Greece (ANIMALBASE). All stygobitic Gastropoda are Caenogastropoda except Acroloxus tetensi Kušcer, 1832 from the Dinaric Mountains in Slovenia, and Hydrophrea from New Zealand, which are Heterobranchia/Pulmonata.
Our survey will reliably point to at least 512 known species of troglobic Gastropoda: 368 in Europe, 17 in Africa[7], 31 in former USSR[21], 1 in Iran[8][9][10], two in China[2], 2 in SE Asia[22], four in Australia[12], 10 in New Zealand (A Guide to the Groundwater Invertebrates of New Zealand, 2003), 39 in USA/Canada[33], 8 in Mexico[32] and 30 in Brazil.

PHYLOGENY OF GASTROPODA, NON-MARINE CLADES IN BRAZIL (FRESHWATER, TERRESTRIAL), AND LINEAGES WITH TROGLOBIC IN BRAZIL
USA/MEXICO
Recent data point to (5:20/)39 spp. for aquatic members in USA/Mexico, by Gladstone et al. (Conservation Biology, 2022). USA has (6:17/)39 spp. of troglobic Gastropoda in Ellobiida/Ellobiidae/Carychiinae (1, former Basommatophora), Hygrophila/Physidae (1, former Basommatophora), Stylommatophora (2:2/4, in Helicodiscus-2 and Glyphyalinia-2), and Littorinimorpha (2:13/33, in Hydrobiidae-8/15 and Cochliopidae-5/18).
EUROPE
There is no exact data on the diversity of troglobic Gastropoda in Western Europe. Here, we will adopt artifically the number of 368, which is the sum of 143 stygobites from France [2], 169 stygobites from the Balkans (Falniowsk, A. et al., Hydrobiologia, 2021), and 56 troglobiontes mentioned in [2]. Europe is home to 11 genera of strictly troglobic snails: Troglaegopis, Sciocochlea, Speleodentorcula, and Spelaeoconcha (all monotypics), Paraegopis (5, 1 TB), Argna (6, 1 TB), Klemmia (2, 1 TB), Cryptazeca (4, 2 TB), Virpazaria (7, 3 TB), Spelaeodiscus (10, 9 TB) and Zospeum (35 TB), mainly in the Dinaric karst.
IN BRAZIL
Cavallaria et al. (Zoologia, 2022) cites 19 described troglobic gastropoda from Brazil, however, troglobic status for many of them are rejected by several autors. Here we accept 28 names as troglobic in Brazil, mentioned expressly in R.L.Ferreira et al. (6, Zoologia, 2023), Salvador & Bichuette (2, Zoosystematics and Evolution, 2024), and 22 not yet described, being 4 in Pará [PA/2], 1 in Rio Grande do Norte [RN/1], 8 in Bahia [BA/2][BA/3][BA/4][BA/7], 2 in Mato Grosso do Sul [MS/1], 6 Potamolithus in São Paulo [SP/1], and 1 in Paraná [PR/1]. Overall, Brazil has at least nine genera and nine families.
Brazilian non-marine gastropods are distributed across eight lineages, with four in each environment. Below, we present information on each of them, as well as on the existence of troglobic lineages in Brazil within each group.
NERITOMORPHA — (2/)34 spp. in Brazil, all terrestrial, none as troglobic in country.
HETEROBRANCHIA
HYGROPHILA — freshwater. It includes six families and (21/)62 spp. in Brazilian freshwater environments, with no troglobic representatives in the country.
STYLOMMATOPHORA — terrestrial, very diverse in Brazil.
CHAROPIDAE


GASTROCOPTIDAE


SCOLODONTIDAE






SYSTELLOMATOPHORA — includes only Veronicellidae in Brazil, true slugs, none known as troglobic in country.
CAENOGASTROPODA
CYCLOPHOROIDEA — terrestrial. Includes (7/)22 spp. in Brazil in three families, one with troglobic in country.
DIPLOMMATINIDAE


AMPULARIOIDEA — freshwater. It includes (5/)49 spp. in Brazil, all belonging to the family Ampullariidae, with no troglobic representatives in the country.
CERITHIOIDEA — freshwater. It includes (2/)32 spp. in Brazil, all belonging to the family Tiaridae, with no troglobic representatives in the country.
TRUNCATELLOIDEA — freshwater. It includes three families in Brazilian freshwater environments, all of which have troglobic species, being the unique lineage with stygobitic species in Brazil.
COCHLIOPIDAE




TATEIDAE







TOMICHIDAE




UNDETERMINED FAMILIES








8 PHYLUM NEMATODA ‣ despite the extreme diversity of habitats they can occupy, in contrast with their marked morphological homogeneity, there is no systematic body of knowledge regarding troglobic Nematoda. Even basic criteria to define whether a species is troglobic do not exist. Although there are numerous collections, there is little precision about what truly constitutes a troglobic Nematoda. A careful review of Du Preez (Nematology, 2017) reveals that seven species are possibly true troglobic: Desmoscolex aquaedulcis Stammer, 1935 (Slovenia), Thalassoalaimus aquaedulcis Schneider, 1940 (Slovenia), Halalaimus stammeri Schneider, 1940 (Slovenia), Hemicycliophora aquatica Loos, 1948 (Belgium), Stenonchulus troglodytes Schneider, 1940 (Slovenia, Austria), Mylonchulus cavensis Schneider, 1940 (Hungary), and Chronogaster troglodytes Poinar & Sarbu, 1994 (Romania). Axonchium sbordonii Zullini, 1973 (Belondiridae), from Sima del Ojito, Chiapas, Mexico, is reported as troglobic from Mexico. In addition, one possibly troglobic species in Rhabditida was collected in Colombia[19]. Additionally, two troglobic Nematoda are known from USA[33]: Amphibiocapillaria texensis Moravec & Huffman, 2000 (Capillariidae, from Texas, parasitic in Typhlomolge rathbuni Stejneger, Caudata, Folia Parasitologica, 1988) and Rhabdochona longleyi Moravec & Huffman, 1988 (Thelaziidae, parasitic on Trogloglanis pattersoni and Satan eurystomus, both Ictaluridae, from the subterranean waters of Texas, Folia Parasitologica, 2000). Thus, a total of 11 troglobic Nematoda are recognized in this text.
9 PHYLUM ONYCHOPHORA ‣ only two troglobic Onychophora species are described worldwide: Peripatopsis alba Lawrence, 1931 (Peripatopsidae) from South Africa, and Speleoperipatus spelaeus Peck, 1975 (Peripatidae) from Jamaica[24] (Subterranean Biology, 2025). There is also a troglobic species in Brazil, not yet formally described, found in Dente do Cão cave of the Bodoquena System, Mato Grosso do Sul state, which may even represent a new genus [MS/1] — unfortunately, not as depigmented as its already described counterparts.
PERIPATIDAE


10A ARTHROPODA/ARACHNIDA ‣ all orders of Archnida has troglobic except Holothrida and Solifugae. 337 troglobic Arachnida occur in North America (in Acari, Pseudoscorpiones, Araneae, Opiliones, Schizomida, Scorpionida)[33], 144 in China[2], 184 in Mexico[32], and 178 in Brazil.
Among Acari, two superorders and four orders contain troglobic species: Parasitiformes with the orders Opilioacarida and Gamasida, and Acariformes with the orders Sarcoptiformes and Trombidiformes. At least five Acari are troglobic in Western Europe, in Belgium (4, SEE) and Italy (4, SEE | SEE), and 4 in Russia [21], 12 in Cuba [13], 1 in Bolivia [15], (10:17/)26 in USA/Canada [2 | 13 in Rhagidiidae], 9 in Venezuela [17], 2 in Mexico [18], 4 in SE Asia [22], and 2 in America Central [23].
In total, 15 mites considered troglobitic could not be assigned to a specific group: 4 in Belgium (SEE), 9 in Australia, 1 in Argentina (ICS/vol VII, 2025), and 1 aquatic mite in Cuba[13].
1 OPILIOACARIDA
Although several species in Brazil are associated with subterranean habitats, none are classified as troglobic (Berbardi & Borges-Filho, Subterranean Biology, 2018). Troglobic worldwide occur SE Asia[21], in Thailand (Siamacarus-2, Vanderhammenacarus-1) and Vietnam (Siamacarus-1).
2 IXODIDA
Galán & Herrera (BSVE, 2006) lists Antricola silvai Cerny, 1967, from Falcon state, as a potential troglobic. 11 troglobic are listed from Cuba[13]. If confirmed, these species are the only record of an Ixodida troglobic worldwide. Huge populations occur in caves in Brazil, associated with bats, all troglophiles.
3 MESOSTIGMATA
In Mesostigmata, a large number of cave species are permanent or temporary ectoparasites of bats, but most are free living. Three troglobic undescribed species are known in Brazil, collected in Minas Gerais [MG/1] and Bahia [BA/3] states. USA has 5 species of troglobic Mesostigmata in Laelapidae (1/1, Kentucky), Macrochelidae (1/2, Kentucky) and Veigaiidae (1/2, Indiana)[33].
UNDETERMINATED FAMILY


DITHINOZERCONIDAE


PACHYLAEPIDAE


4 TROBIDIFORMES
Trombidiformes exhibits the most remarkable diversification of troglobitic species among all Acari groups, often representing the entirety of such fauna in several regions worldwide, with species also recorded in Madagascar (1, Neotrombidiidae)[7], Italy and Austria (Rhagidiidae, Journal of Natural History, 2011 | NDVI, 1994), Russia (7, Rhagidiidae, Rhagidia-4, Foveacheles-1)[21], SE Asia (3, Eutrombidiidae-1, Leeuwenhoekiidae-2)[22], New Zealand (71, all Hydracaria), USA/Canada (25, Aturidae-1/1, Florida and Texas; Bogatiidae-1/1, Illinois; Cocceupodidae-1/1, Kansas; Hydryphantidae-1/1, Texas; Hygrobatidae-1/3; Limnesiidae-1/1, Texas; Limnohalacaridae-2/3, all in Indiana; Nudomideopsidae-1/1, Texas; Rhagidiidae-7/13, widely distributed)[33], Mexico (Proterorhagia oztotloica Lindquist and Palacios-Vargas, 1991, Proterorhagiidae; and Robustocheles infernalis Zacharda & Elliott, 1985, Rhagidiidae)[32], Venezuela (3:8, all Hydracnidia)[17], Bolivia (1, Rhagidiidae)[15] and Brazil (5:5, all undescribed).
Prostigmata includes a few poorly known cave species in the families Leeuwenhoekiidae, Trombiculidae, and Trombidiidae, frequent on or close to guano accumulations intropical caves, and bat or invertebrate parasites at larval stage. Proterorhagidiidae has a single relictual troglobic, Proterorhagia oztotloica Lindquist & Palacios-Vargas, 1991 from Mexico. In Brazil occur a single described and four undescribed species in Pará [PA/2], Bahia [BA/1] and Minas Gerais [MG/4] states.
UNDETERMINATED FAMILY


ERYTHRAEIDAE


LABIDOSTOMATIDAE


RHAGIDIIDAE


SPHAEROLICHIDAE


5 SARCOPTIFORMES (ORIBATIDA)
Troglobic species of this group are surprisingly rare: some species of Schwiebea (Acaridida) from humid hypogean habitats, devoid of adaptations to subterranean life, and a few European Belbidae, such as Metabelbella phalangioides are pale and long-legged[4]. Brazil has two undescrib species in Bahia [BA/3] and Mato Grosso do Sul [MS/1] states. USA has two species of troglobic Sarcoptiformes, both from Kentucky: Belba bulbipedata (Packard, 1888) in Belbidae, and Galumna alata (Hermann, 1804) in Galumnidae[33].
HYDROZETIDAE


OEHSERCHESTIDAE


6 PSEUDOSCORPIONES
A total of 460 troglobic Pseudoscorpiones species worldwide in 15 families are included in our survey, being 132 in Balkans[29], 7 in Africa[7], 7 in Russia and adjacent countries[21], 40 in China[2], 7 in Korea (Plos One, 2025), 5 in SE Asia to New Guines[22], 6 in Australia, (8:27/)153 in USA/Canada (Bochicidae-1/1, Texas; Chernetidae-6/12; Chthoniidae-7/97, mainly in Alabama; Ideoroncidae-1/1, Arizona; Larcidae-1/5, California, Arizona and Texas; Neobisiidae-8/27, mainly in Texas; Pseudogarypidae-1/3, Arizona and California; Syarinidae-2/7)[33], (5:)40 in Mexico[32], 3 in Cuba[13], 4 in Jamaica[24], 1 in Guatemala[31], 1 in Aruba (Zoologia, 2023), (6:)53 in Brazil and 1 in Argentina.
21 undescribed troglobic species occur in Brazil, in Pará [PA/2], Rio Grande do Norte [RN/1], Bahia [BA/2][BA/3][BA/4], Minas Gerais [MG/1][MG/6][MG/10], Goiás [GO/1], São Paulo [SP/3], Paraná [PR/1] states.
BOCHICHIDAE






CHTHONIIDAE






























CHERNETIDAE














IDEORONCIDAE


OLPIIDAE


SYARINIDAE


7 ARANEAE
648 troglobic species of Araneae worldwide in 48 families: 194 in Europe (Scientific Data, 2022), 16 in Africa[7], 2 in Turkmenistan (WHC Unesco), 1 in Iran[8][9][10], (16:30/)104 in China [2], 36 in SE Asia to New Guinea[21], 13 in Australia, (8:23/)121 in USA/Canada (Dictynidae-52, all in Cicurina from Texas except one in Alabama; Leptonetidae-5/25, mainly in Texas; Linyphiidae-9/15, widely distributed; Lycosidae-2/2, both in Hawaii; Nesticidae-2/21, mainly in Tenessee and Texas; Ochyroceratidae-2/2, California and Hawaii; Tengellidae-2/3; Theridiidae-1/1, Arizona)[33],(11:)58 in Mexico[32], 7 in Cuba[13], 9 in Jamaica[24], 1 in Guatemala[31], 1 in Trinidad[23], 3 in Colombia (three families, Pisauridae without Brazilian troglobic)[19], 79 in Brazil and 1 in Argentina.
Brazil has (16:)79 spp., 44 of thesse undescribed in Pará [PA/2], Tocantins [TO/1], Rio Grande do Norte [RN/1], Bahia [BA/1][BA/2][BA/3][BA/4][BA/5][BA/6][BA/7], Minas Gerais [MG/1][MG/3][MG/5][MG/10], Espírito Santo [ES/1], São Paulo [SP/2], Mato Grosso do Sul [MS/1] and Paraná [PR/1] states.
Among Theraphosidae, troglobic species includes Orphnaecus pellitus Simon, 1892 from Philippines (Subterranean Biology, 2025), six Hemirrhagus from Mexico[32], and Tmesiphantes hypogaeus Bertani, Bichuette & Pedroso, 2013 from Brazil (Bertane, Bichuette & Pedroso, Anais da Acadêmia Brasileira de Ciências, 2013).
UNDETERMINATED FAMILY




CAPONIIDAE






CTENIIDAE


DIPLURIDAE


HAHNIIDAE


MICROSTIGMATIDAE


NESTICIDAE


OCHYROCERATIDAE





















OONOPIDAE








PALPIMANIDAE


PHOLCIDAE




PRODIDOMIDAE














SICARIIDAE




SYMPHYTOGNATHIDAE


TELEMIDAE


TETRABLEMMIDAE








THERAPHOSIDAE


8 OPILIONES
Worldwide there are 159 known troglobic Opiliones, in Balkans in Europe (21)[29], South Africa (2)[7], Russia and adjacent countries (8)[21], SE Asia to New Guinea (3)[22], 2 in Australia, USA (7:12/46, in Cladonychiidae-1/2, Idaho; Paranonychidae-1/7; Phalangodidae-6/26, mainly Texas and California; Sabaconidae-1/3; Stygnopsidae-1/2, Texas; Taracidae-1/5, California, Oregon and Nevada; Travuniidae-1/1, Washington)[33], Mexico (4:17, Journal of Natural History, 2025)[32], Cuba (4)[13], Jamaica (2)[24], Guatemala (1)[31], Venezuela (4, all in Trinella in Agoristenidae)[17], Colombia (3)[19], Brazil (44, all Laniatores) and Argentina (1).
Four suborders comprises this order. In Cyphophthalmi the unique New World troglobic is Neogovea mexasca Shear, 1977 from Mexico. Eupnoi no has true troglobic. Dyspnoi (Palpatores) has only one troglobic species in New World, Ortholasma sbordonii Šilhavý, 1973, also in Mexico. All remaining troglobic Opiliones in New World belongs Laniatores[2][4]. Brazil has 28 undescribed troglobic Opiliones, in Pará [PA/1][PA/2], Rio Grande do Norte [RN/1], Bahia [BA/2][BA/3][BA/7], Espírito Santo [ES/1], Minas Gerais [MG/1][MG/3][MG/6], Mato Grosso do Sul [MS/1] and Paraná [PR/1] states.
UNDETERMINATED FAMILY




COSMETIDAE


CRYPTOGEOBIIDAE




ESCADABIIDAE














GERDESIIDAE


GONYLEPTIDAE


















KIMULUDAE


TRICOMMATIDAE


ZALMOXIDAE


9 AMBLYPYGI
Troglobic in Amblypygi (25 species worldwide), are cited in four families, three in New World: Charinidae (Charinus in New World, including all 11 Brazilian troglobic and two in Venezuela[17], without troglobic in Mexico; Paracharontidae, with Jorottui, a monogeneric troglobic endemic to N Colombia (Novataxa, 2023); and Phrynidae, with true troglobic in Mexico (6)[32], Cuba (1)[13] and Colombia (1)[19]. No troglobic Amblypygi occur in USA. Worldwide, troglobic occur also in Guinea (Charinus milloti Fage, 1939)[7], Vietnam (Diversity, 2023) and Sulawesi (Diversity, 2021).
CHARINIDAE











10 SCHIZOMIDA
Both two families of this order has troglobic. Troglobic Hubbardiidae known from caves in Tanzania (Trithyreus cavernicola Hansen, 1926)[7], Sulawesi (1, Diversity, 2021), Australia (1), USA/Canada (1, Hubbardia shoshonensis Briggs & Hom, 1972, from California)[33], Mexico (12)[32], Cuba (5)[13], Jamaica (1)[24], Ecuador (1) and Brazil, with a single undescribed spcies from Pará state [PA/2]. In Protoschizomidae, all 10 spp. are troglobic, nine in Mexico[32] and Agastoschizomus texanus Monjarez-Ruedas et al., 2016 in Texas, USA[33].
HUBBARDIIDAE


11 THELIPHONIDA
A single troglobic is known worldwide, Typopeltis magnificus Haupt, 2004, from Laos[4][22].
12 PALPIGRADI
62 troglobic Palpigrada are known worldwide, in Europe (23, Forschung), Abkhazia (1)[21], Indonesia (1, Prokoenenia celebica)[22], Australia (1, Eukoenenia guzikae, Invertebrate Systematics, 2008), Cuba (1)[13], 33 in Brazil, and 2 in Argentina.
All genera in this order except Leptokoenenia have troglobic[2]. In New World, troglobic Palpigrada occur only in Brazil (Allokoenenia-2, Eukoenenia-28, three undeterminate at genus-level), Cuba and Argentina. 12 undescribed in Brazil, in Bahia [BA/1][BA/2][BA/3][BA/6][BA/7], Minas Gerais [MG/1], São Paulo [SP/2] and Paraná [PR/1] states.
EUKOENENIDAE


































13 RICINULEI
Troglobic Rcinulei in Pseudocellus have been described from Mexico (11) and Cuba (4), and Cryptocellus armasi in known troglobic from NW Venezuela (1, Zootaxa, 2025). Troglobic Ricinulei does not occur in Brazil.
14 SCORPIONIDA
There are 38 troglobic scorpions worldwide, in Madagascar (2, Zoosystema, 2008), Montenegro (1, Biologia Serbica, 2020), Israel (1, Akrav israchanani, Zoology in the Middle East, 2007), Indonesia (1, Invertebrate Systematics, 2008), Malaysia (1, Invertebrate Systematics, 2008), Laos[22], Vietnam[22], Australia (2, Invertebrate Systematics, 2008 | British Arachnological Society, 2021), USA in California (1, Uroctonus grahami Gertsch & Soleglad, 1972, Vaejovidae)[33], Mexico (16)[32], Cuba (3, Euscorpius, 2021), Bahamas (1, Euscorpius, 2021), Dominican Republic (2, Euscorpius, 2021), Venezuela (Taurepania trezzii Vignoli & Kovařík, 2003, Chactidae)[19], Ecuador (1, Troglotayosicus vachoni Lourenço, 1981, Troglotayosicidae), and Brazil (3, Buthidae, all in Bahia state).
BUTHIDAE




10B CHILOPODA ‣ of the five orders of Chilopoda, four have cave species, all of them in Brazil. For cave Chilopoda worldwide, see Chagas and Bichuette (Zookeys, 2018). 55 troglobic occur in Europe (50 Lithobiidae, Cryptopidae in Spain and Italy, and Geophilidae in France and Croatia)[29], 3 in Africa (in Lithobiomorpha and Scolopendromorpha)[7], 2 in Russia and adjacent former Soviet republics[21], 1 in China[2], 2 in Australia, 4 in USA (Lithobiomorpha and Cryptopidae)[33], (2:)5 in Mexico (two Lithobiidae, 3 in Newportia)[32], 1 in Panama[23], 1 in Caribbean, 1 in Colombia[19], and 21 in Brazil (11 Scolopendromorpha, 7 Geophilomorpha, two Lithobiomorpha, one Scutigeromorpha). All six families with troglobic has species in Brazilian caves except Balophilidae[4], and Lithobiidae (if Brazilian Lithobiomorpha members all belong to Henicopidae).
1 SCUTIGEROMORPHA
Sphendononema sp. collected in Andaraí municipality, center Bahia state [BA/3] in Brazil representes the unique troglobic Scutigeromorpha worldwide.
PSELLIOLIDAE


2 LITHOBIOMORPHA
About 50 troglobic species of this order (all Lithobiidae) occur in many countries in Europe[2], one in Algeria [7], two in North America (Nampabius turbator Crabill, 1952 from Virginia and Stenophilus californicus (Chamberlin, 1930) from California)[33], two in Mexico[32], and remaining from E Asia. Two undescribed species was collected in Rio Grande do Norte [RN/1] and Bahia [BA/2] states in Brazil.


3 SCOLOPENDROMORPHA
Five families comprise this order, three of them has troglobic. Scolopendridae includes Otostigmus cooperi (Panama) and one species in Tanzania; Cryptopidae includes Theatops phanus Chamberlin, 1951 (Texas, USA), Thalkethops grallathrix Crabill, 1960 (New Mexico, USA), Cryptops longicornis (Spain), C. vulcanicus (Canaries, Spain), C. umbricus (Italy), Cryptops legagus (Botswana), C. roeplainsensis, C. camoowealensis (Australia), C cavernicolus, C. troglobius (Cuba), C. iporangensis, C. didi and C. spelaeoraptor (Brazil); and Scolocryptopidae includes Newportia leptotarsis (Cuba), three in Mexico, N. stoevi (Puerto Rico), N. spelaea, N. potiguar, N. eleonorae, and Scolopocryptops troglocaudatus in Brazil, and Scolocryptops sp. in Colombia[9]. Three undescribed species occur in Brazil in Cryptopidae, in Pará [PA/2] and Bahia states [BA/2][BA/3], and 4 in Scolocryptopidae, in Rio Grande do Norte state [RN/1].
CRYPTOPIDAE






SCOLOCRYPTOPIDAE






4 GEOPHILOMORPHA
11 troglobic Geophilomorpha species are known worldwide, of which 5 have been formally described (Nunes et al., Zootaxa, 2019): Plutogeophilus jurupariquibara (Brazil), Geophilus persephones (France) and G. hadesi (Croatia) in Geophilidae; Ityphylus cavernicolus (Cuba) in Ballophilidae; and Schendylops janelao (known only from nortern Minas Gerais state, Brazil) in Schendylidae. Five undescribed species was collected in Brazil, in Bahia [BA/1][BA/2], São Paulo [SP/3], Rio Grande do Norte [RN/1] and Minas Gerais [MG/5] states.
GEOPHILOMORPHA UNDETERMINATED FAMILY




GEOPHILIDAE


SCHEDYLIDAE


10C SYMPHYLA ‣ only nine species of this order are described as troglobic: 1 in Tasmania (Hanseniella magna Scheller, 1996, Australian Journal of Entomology, 1996), 2 in Slovenia, 1 in Argentina (ICS/vol VII, 2025), and 5 in Brazil, all these undescribed — one in the Alto do Ribeira karst, São Paulo state [SP/3], 3 in Minas Gerais [MG/6], and one in Padre Cave, Bahia [BA/7]. If confirmed as troglobic, they would represent the first Symphyla troglobic described from the New World.






10D PAUROPODA ‣ no described troglobic Pauropoda is currently known[2] — however, one undescribed species was collected in Minas Gerais state [MG/1], SE Brazil. If confirmed, it would be the first troglobic Pauropoda species described worldwide.


10E DIPLOPODA ‣ our survey documented indicate 672 troglobic Diplopoda species worldwide, although this estimate is highly imprecise — particularly due to the uneven sampling effort and high diversity in Europe, also for Japana and Australia. There are 191 in Europe [29 | Iberian Peninsula | Romania], 12 in Russia and adjacent former Soviet republics[21], 4 in Africa[7], 1 in Iran[8][9][10], 114 in China[2], 15 from SE Asia to New Guinea[22], 9 in Australia, 140 in the USA and Canada[33], 73 in Mexico[5], 2 in Guatemala[23][31], 1 in Venezuela[17], 6 in Colombia[19], 96 in Brazil (in 5 orders and at least 15 families, 81 undescribed), and 1 in Argentina (Arthropoda, 2025). Troglobionts are known in 12 of the 16 orders of Diplopoda.
UNDETERMINATE ORDERS


1 POLYXENIDA
Possibly nine troglobic known worldwide, three described (Lophoproctus pagesi Condé, 1981 from Balearic Islands, Lophoturus speophilus Nguyen Duy-Jacquemin, 2014, and L. humphreysi Nguyen Duy-Jacaquemin, 2014 from Christmas Island)[2] and six undescribed, all in Brazil, in Pará [PA/2], Bahia [BA/4] and Minas Gerais [MG/7] states.
HYPOXEGENIDAE


LOPHOPROCTIDAE




POLYXENIDAE


2 GLOMERIDA
Troglobic in this order occur in Europe, China, two in SE Asia[22], Mexico (six microphthalmic troglobic in Glomeroides)[5], and America Central. 26 troglobic are collected in Old World.
3 SIPHONOPHORIDA
Possibly only four troglobic worldwide: one in Mexico, one in Texas (Siphonophora texascolens Chamberlin & Mulaik, 1941)[33], one in California, USA (Illacme tobini Marek et al., 2016)[33], and one undescribed in Bahia state [BA/5], NE Brazil.
SIPHONOPHORIDAE


4 GLOMERIDESMIDA
A smaller order, with only five troglobic species, all in Glomeridesmus, G. sbordonii Shear, 1974 from Mexico[2][4], and four in N Brazil, G. spelaeus Iniesta & Wesener, 2012 and three undescribed [PA/1][PA/2], all from Pará state, in N Brazil (Iniesta, Zootaxa, 2012 | M.Souza-Silva et al., Journal of Insect Biodiversity, 2025).
GLOMERIDESMIDAE




5 JULIDA
Dominant millipede order in Europe and many troglobic have been described. There are two troglobic species in USA (both Zosteractinidae, from Alabama and Georgia)[33], but relatively few in Asia, and none are known from Mexico[2][4].
6 SPIROBOLIDA
This order has at least three troglobic: Reddellobus troglobius Causey, 1975 in Mexico, Speleostrophus nesiotes Hoffman, 1994, depigmented and eyeless, in Australia[2][4], and one undescribed in Colombia (Rhinocricidae)[19].
7 CALLIPODIDA
Troglobic species in this order are found in USA (two Tetracion, Abacionidae, from Alabama and Tennessee)[33], China, and 7 in SE Asia[22]. Unknown in Mexico[2][4].
8 SPIROSTREPTIDA
This order is subdivided into Cambalidea and Spirostreptidea. Cambalidea includes 8 troglobic species in New World: Cambala speobia Chamberlin, 1953 from gypsite caves in Texas (USA)[33], and (2:3/)7 in Mexico (Iniesta & Ferreira, Zootaxa, 2013). One troglobic occur in Africa[7], one in Iran[9] (Cambalidae) and one in Colombia[19] (Spirostreptida). In Brazil all troglobitc are Pseudonannolene (Spirostreptidea) except one Spirostreptidae from Serra dos Carajas. Six undescribed species occur in caves from Pará [PA/2], Bahia [BA/2], Espírito Santo [ES/1] and Goiás [GO/1] states.
PSEUDONANNOLENIDAE








SPIROSTREPTIDAE


9 CHORDEUMATIDA
The most important millipede order in north temperate zone caves is the Chordeumatida. USA has (3:8/)108 spp., in Caseyidae (2/3, California and Oregon), Cleidogonidae (62, all in Pseudotremia, widely distributed), Conotylidae (7/12), Striariidae (2/3, California), Tingupidae (2/2, Colorado and Illinois) and Trichopetalidae (6/26, mainly Alabama and Arkansas)[33]. One troglobic occur in Africa[7]. (2:2/)13 troglobic in Mexico[5]. Heterocaucaseuma deprofundum Antic & Reboleira, 2018, is a troglobic from deep caves in the Abkhazia, and at 1,980m below the surface, is the deepest occurring known millipede and perhaps the deepest occurring terrestrial arthropod worldwide (Zootaxa, 2018).
10 STEMMIULIDA
Besides some cave-dwelling species (some Stemmiulus in N Brazil), possibly only one species is assignated as troglobic, from Colombia[19].
11 POLYDESMIDA
75 troglobic species in Brazil, 11 and 64 undescribed, in Pará [PA/1][PA/2], Rio Grande do Norte [RN/1], Bahia [BA/1][BA/2][BA/3][BA/4][BA/6][BA/6], Minas Gerais [MG/1][MG/3][MG/5][MG/6][MG/10], Espírito Santo [ES/1], São Paulo [SP/2][SP/3][SP/4], Paraná [PR/3], Goiás [GO/1] and Mato Grosso do Sul [MS/1]. (3:8/)25 spp. troglobic species in USA, in Macrosternodesmidae (6/18, mainly in W North America), Nearctodesmidae (1/1, Ergodesmus remingtoni (Hoffman, 1962), from Illinois) and Polydesmidae (1/6, all from Texas)[33].
UNPLACED AT FAMILY LEVEL










APHELIDESMIDAE


CHELODESMIDAE












CRYPTODESMIDAE






DOBRODESMIDAE


ONISCODESMIDAE











PARADOXOSOMATIDAE










PYRGODESMIDAE
















SPHAERODESMIDAE


TRICHOPOLYDESMIDAE (INCLUDES ALL RECORDS FOR FUHRMANNODESMIDAE)









10F OLIGOSTRACA ‣ although information is limited, it is estimated that approximately 600 species are stygobitic[2] Ostracoda, however our numbers indicate only 232, being 114 in Europe[30], 4 in Africa[7], 5 in Russia and adjacent countries[21], 74 in Australia[12], (2:14/)22 in the USA (all Podocopida, highly centered in Texas)[33], (2:2/)2 in Mexico (excludes anchialine Danielopolina mexicana Kornicker & Iliffe)[5], 5 in Cuba[13], 3 in Jamaica[24], 2 in Venezuela (Cyprididae)[17], and one in Brazil, a undescribed species collected in Rio Grande do Norte [RN/1] state. None from China.


10G MALACOSTRACA ‣ 7 of 17 orders has obligate-cave species in terrestrial and freshwater environments. Brazil and China has troglobic in Amphipoda, Bathynelllacea, Isopoda and Decapoda. Brazil also has in Spelaeogriphacea. 379 obligate subterranean species in North America[33], 68 in China[2], 99 in Mexico[32] and 177 in Brazil.

PHYLOGENY OF MALACOSTRACA (BRAZILIAN METAZOA) AND TROGLOBIC IN BRAZIL IN GREEN
1 BATHYNELLACEA
177 spp. of Bathynellacea are troglobic worldwide: 106 in Europa[30], 27 in Africa[7], 15 in Russia and adjacent countries[21], 1 in China[1], 1 in Australia, 5 in New Zealand, (2:7/)23 in USA (mainly California and Texas)[33], 1 in Venezuela (Parabathynellidae)[17], and 1 in Brazil, a undescribed species collected in Mato Grosso do Sul state [MS/1].


2 ANASPIDACEA
Five families from Australia, New Zealand and southern South America. All members in Psammaspididae (1, Australia), Stygocarididae (8, Australia 1, New Zealand 2, Chile 2, Argentina 3) and Patagonspididae (1, Argentina) are stygobitic.
3 AMPHIPODA
1,021 troglobic worldwide documented in Amphipoda this article: 438 in Europe[30], 81 in Africa[7], 43 in Russia and adjacent countries[21], 24 in Iran[8][9][10], (5/)26 in China[2], 3 in Korea (PeerJ), 5 in SE Asia to New Guinea[22], 140 in Australia[12], 4 in New Zealand, (11:24/)192 in USA/Canada[33], (2:)22 in Mexico (Bogidiellidae and Hadziidae)[32], 3 in Cuba[13], 4 in Jamaica[24], 3 in smaller islands of Caribbean[23], (2:)2 in Venezuela (Bogidiellidae and Hyalellidae)[17], 30 in Brazil and 2 in Argentina.
(4:5/)30 in Brazil, in Hyalella (9), Megagidiella (2), Potiberaba (5, VIDEO), Spelaeogammarus (11) and Seborgia (1). Five undescribed species in Brazil: 4 collected in Rio Grande do Norte [RN/1] and 1 in Mato Grosso do Sul [MS/1] states.
USA has (11:24/)192 troglobic Amphipoda, in Aoridae (1/1, Hawaii), Artesiidae (1/2, Texas), Brevitalitridae (1/2, Hawaii), Crangonyctidae (5/165, 139 in Stygobromus), Eusiridae (1/3, Hawaii), Gammaridae (1/3, Illinois, Pensylvania, Virginia and West Virginia), Hadziidae (8/8, Hawaii and Texas), Hyalellidae (1/1, Nevada), Melitidae (2/3, Hawaii), Parabogidiellidae (2/2, Texas), and Seborgiidae (1/2, Texas)[33].

PHYLOGENY OF AMPHIPODA (WIKIPEDIA | ZOOTAXA 2013, PG18) AND TROGLOBIC IN BRAZIL
AMPHILOCHILOIDEA/SEBORGIIDAE




SENTICAUDATA/BOGIDIELLIDA/ARTESIIDAE












SENTICAUDATA/BOGIDIELLIDA/BOGIDIELLIDAE


SENTICAUDATA/TALITRIDA/HYALELLIDAE









SENTICAUDATA/GAMMARIDA/MESOGAMMARIDAE






4 INGOLFIELLIDA
A single troglobic reported worldwide, Proleleupia nudicarpus Griffiths, 1991, endemic to Namibia[7].
5 THERMOSBAENACEA
(4:8/)45 spp. worldwide (Catalogue of Life | Brazilian Metazoa), one of the most enigmatic and challenging for determining troglobic status. Many of the 45 known species inhabit wells, cave lakes, mines, cenotes, interstitial environments, cold and hot springs, and aquifers with salinity ranging from oligohaline to polyhaline, as well as marine and anchialine caves in various regions of the world (H.P.Wagner, Monography, 1994, and recent described species). A careful and detailed analysis allows only ten species to be considered truly troglobic in freshwater environments, in Cuba (1, Tethysbaena vinabayesi), Puerto Rico (1, T. colubrae), USA (2, Tethysbaena texana and Salishbaena kootenai), France (1, T. exigua), Italy (T. argentarii), Thailand (2), Cambodia (1) and W Australia (1, Halosbaena tulki).
6 SPELAEOGRIPHACEA
A fully freshwater order with (1:3/)4 spp. in Spelaeogriphidae[2], all troglobic, in WC Brazil (Potiicoara), NW Australia (Mangkurtu), and South Africa (Spelaeogriphus). Brazilian member is troglobic Potiicoara brasiliensis Pires, 1987, known only caves from Lagoa Azul in Bonito municipality, Gruta Ricardo Franco near Corumbá municipality, and Gruta do Curupira in the Araras Mountains, Mato Grosso state (Vanin, Zootaxa, 2012).
Recent genetic studies have investigated whether Potiicoara brasiliensis is indeed a single species or rather a species complex, given its controversial distribution for a troglobic taxon (ICMBIO/News).


7 DECAPODA
Worldwide, 188 species of Decapoda occur only in subterranean environments, in four infraorders. Caridea includes a huge of shrimps. Astacidae includes more than 40 stygobitic species of crayfishes of different genera and in environments of temperate and tropical freshwater caves, for Papua New Guinea, Cuba, Mexico, and North America. Anomura includes troglobic only in SE Brazil (and marine forms in Canary Is.). Brachyura has troglobic crabs in Pseudothelphusidae and Trichodactylidae[2].
Troglobic Decapoda includes 16 in Europe[30], 15 in Africa[7], 7 in Russia and adjacent countries[21], 23 in China[2], 27 in SE Asia to New Guinea[22], 3 in Australia, 44 in USA/Canada[33], 26 in Mexico[32], 13 in Cuba[13], 2 in Jamaica[24], 3 in América Central (Guatemala and Belize, SEE), 1 in Puerto Rico[23], 2 in Venezuela (Chaceus caecus and Eudaniella sp.)[17], and six in Brazil.
Mexico has 20 spp. of troglobic freshwater Decapoda, in Alpheidae (shrimp, 1, Potamalphaeops stygicola), Palaemonidae (shrimp, 9), Cambaridae (crayfishes, 5), Pseudothelphusidae (crabs, 4) and Trichodactylidae (crabs, 1). USA has (3:7/)44 spp.[33], in Procarididae (shrimps, 2/2, both in Hawaii), Cambaridae (5/35, crayfishes, mainly in Alabama, Florida and Tennessee), Atyidae (shrimps, 4, Palaemonias in Kentucky and Alabama, and Halocaridina in Hawaii), and Palaemonidae (shrimps, 2/3, Texas and Florida).
A conspectus of the diversity of troglobic Aegla in Brazil is available in the Proceedings of the 19th International Congress of Speleology, vol. VI/P99.
AEGLIDAE





8 ISOPODA
This order, along with Collembola, Amphipoda, Araneae and Coleoptera, are among the major groups in terms of troglobic diversity worldwide. Our survey documented 880 troglobic species, including 337 in Europe[30], 29 in Russia and adjacent countries[21], 84 in Africa[7], 3 in Iran[8][9][10], 18 in China[2], 29 in SE Asia to New Guinea[22], 5 in Australia, 4 in New Zealand, (7:21/)118 in USA/Canada (Asellidae-11/90, 56 in Caecidotea; Cirolanidae-3/4, Texas and Virginia; Ligiidae-1/1, California; Microcerberidae-1/3, Texas; Philosciidae-1/4, Hawaii; Stenasellidae-1/2, Florida and Texas; and Trichoniscidae-3/14)[33], (11:)52 in Mexico[32], 7 in Cuba[13], 4 in Jamaica[24], 2 in Guatemala[31], 3 in other Caribbean islands[23], 5 in Venezuela (Calabozoidae, Cirolanidae, Sphaeroniscidae, Oniscidae and Pillosciidae)[17], 2 in Colombia (Armadillidium sp.[19] and Diploexochus cacique), 1 in Peru[16], 1 in Bolivia[15], 140 in Brazil and 4 in Argentina.
(12:)140 in Brazil (at least 28 genera), 64 undescribed, in Pará [PA/1][PA/2], Tocantins [TO/1], Rio Grande do Norte [RN/1], Minas Gerais [MG/1][MG/4][MG/6][MG/10], Bahia [BA/1][BA/4][BA/6][BA/7], Espirito Santo [ES/1], São Paulo [SP/2], Paraná [PR/1], Goias [GO/1] and Mato Grosso do Sul [MS/1].
CYMOTHOIDA/CIROLANIDAE






CALABOZOIDA/BRASILEIRINIDAE


CALABOZOIDA/CALABOZOIDAE








ONISCIDEA UNPLACED FAMILY


ONISCIDEA/SYNOCHETA/STYLONISCIDAE (VIDEO)
Chaimowiczia




Cylindroniscus


Iuiuniscus


Pctenoniscus




















Spelunconiscus


Xangoniscus


















STYLONISCIDAE UNDETERMINATED GENUS




















ONISCIDEA/CRINOCHETA UNPLACED FAMILY


ONISCIDEA/CRINOCHETA/ARMADILLIDAE



ONISCIDEA/CRINOCHETA/BALLONISCIDAE




ONISCIDEA/CRINOCHETA/DUBIONISCIDAE


ONISCIDEA/CRINOCHETA/PHILOSCIIDAE










ONISCIDEA/CRINOCHETA/PLATHYARTHRIDAE






















ONISCIDEA/CRINOCHETA/PUDEONISCIDAE






ONISCIDEA/CRINOCHETA/SCLEROPACTIDAE














ONISCIDEA/CRINOCHETA/TRACHELYPODIDAE


10H BRANCHIOPODA ‣ (1:2/)7 troglobic species are knonw in this order, all in Chydoridae from Europe, in Spain, Romania, France, Slovenia and Bosnia & Herzegovina (Hydrobiologia, 1990)[2][30], mainly in Alona.
10I COPEPODA ‣ our survey identifies 814 troglobic copepods (all stygobitic) worldwide, with a explosive radiation of 547 recorded in Europe[30], 67 in Africa[7], 31 in Russia and neighboring countries[21], 12 in Iran[9][10][11], 23 in China[2], 83 in Australia[12], (5:7/)18 in the USA/Canada (15 Cyclopidae, 9 Diacyclops, mainly in Indiana and Tennessee)[33], 10 in Mexico[32], 18 in Cuba[13], 1 in Venezuela (Cyclopidae)[17], and 4 in Brazil, all undescribed, in Rio Grande do Norte [RN/1], Bahia [BA/7], Minas Gerais [MG/6] and São Paulo [SP/3] states.
UNKNOWN GROUP


HARPACTICOIDEAE


CYCLOPOIDEA/ENTOBIIDAE


10J COLLEMBOLA ‣ a total of 733 troglobic Collembola species were recorded in this study, including 338 in Europe (Fiera, C. et al, JZSER, 2021), 4 in Africa (Morocco, Madagascar)[7], 35 in Russia and neighboring countries[21], 42 in China[2], 43 in SE Asia[22], 7 in Australia (see text), (7:16/)101 in the U.S.A and Canada[33], (9:)43 in Mexico[5], 7 in Cuba[13], 2 in Jamaica[24], 1 in America Central[23], 1 in Venezuela[17], 1 in Peru[16], 1 in Bolivia[15], 4 in Argentina (see text) and 103 in Brazil. Undoubtedly, thousands of additional species remain undiscovered in caves around the world.
All orders of Collembola has troglobic in Brazil (Zeppelini, Pre Print, 2021 | G.A.S.Vaz et al., Acta Oecologica, 2024). Onychiurus acuitlapanensis Palacios-Vargas & Deharveng, 1982 (Onychiuridae) is a unique troglobic Collembola from Venezuela, known also from Guerrero state in Mexico (Galán & Herrera, BSVE, 2006).
UNDEFINED ORDER ‣ a species identified in N Minas Gerais [MG/5] cannot be detected at the order level.

SYMPHYPLEONA
Two troglobic species of this order, known from SE Bahia [BA/1][BA/6], cannot be detected at the family level. All troglobic members of this order belongs Arrhopalitidae, Bourletiellidae and Sminthuridae, all present in Brazilian caves[4]. Mexico has 4 troglobic species in two families. USA has 31 spp. in this order, all Pygmarrhopalites (Arrhopalitidae), mainly in Virginia state.
UNDEFINED FAMILY ‣ a species identified in Pará [PA/2]and Bahia [BA/2][BA/6] states cannot be detected at the family level.




ARRHOPALITIDAE ‣ especially diversified in temperate regions of northern Hemisphere, also present in South America and SE Asia. 16 troglobic in Brazil, 8 undescribed, in Rio Grande do Norte state [RN/1], Bahia [BA/2][BA/4], and Minas Gerais [MG/3][MG/10] states.
















BOURLETIELLIDAE ‣ one undescribed troglobic species known in Brazil, in Pará state [PA/2].


SMINTHURIDAE ‣ cave species widely in southern Hemisphere, with Adelphoderia in Australia, Troglospinotheca in Argentina, Troglobentosminthurus in Brazil, and Pararrhopalites widely distributed in the tropics, all poorly known taxonomically. 4 undescribed troglobic species known for Brazil, in Bahia [BA/1] and in Minas Gerais [MG/3] states.










PODUROMORPHA
Troglobic Poduromorpha belong in four of the nine families of Poduromorpha order: Gulgastruridae, Hypogastruridae (unique confirmed with troglobic in Brazil), Onychiuridae, and Neanuridae. Troglobic Hypogastruridae of oligotrophic habitats are often characterized by slender or extremely slender claw, but elongation of appendages is moderate; guanobitics have usually reduced eyes and pigment and sometimes elongated claw like several Pseudacherontides. Three troglobic species are known in Brazil, two of these undescribed, in Bahia [BA/7] and Minas Gerais [MG/2][MG/10] states. Mexico has 11 troglobic species in order, in all four 4 families. USA has (3:7/)20 spp. in Hypogastruridae (3/10, mainly Colorado and Alabama), Neanuridae (3/3, Hawaii and Texas), and Onychiuridae (1/7, widely distributed)[33].
PODUROMORPHA UNDETERMINED FAMILY


HYPOGASTUTIDAE




ENTOMOBRYOMORPHA
Order is constituted by nine extant families, six of them include troglobic and four of these has of them in Brazil (35 undescribed species). Four species of this order known in Pará [PA/2], SE Bahia [BA/1][BA/7] and one in São Paulo [SP/3] cannot be detected at the family level. Mexico has 27 spp. in 4 families. USA has (3:8/)50 spp. in Entomobryidae (5/40), Oncopoduridae (1/7), and Tomoceridae (2/3)[33].
UNPLACED AT FAMILY LEVEL




CYPHODERIDAE ‣ are all phyletically blind and unpigmented, mainly myrmecophilous or termitophilous, but Cyphoderus and Troglobius have troglobic-guanobitic species in tropical caves. None exhibits appendage or claw elongation. 17 troglobic spp. in Brazil, one described and 16 undescribed, in Pará [PA/2], Rio Grande do Norte [RN/1], Minas Gerais [MG/1], São Paulo [SP/2], Mato Grosso do Sul [MS/1] and Goiás [GO/1] states.












ENTOMOBRYIDAE ‣ subterranean radiations are known for many genera, in various regions of the world. 19 troglobic species known in Brazil, nine undescribed, in Rio Grande do Norte [RN/1], Bahia [BA/3], Minas Gerais [MG/3] and Mato Grosso do Sul [MS/1] states.


















ISOTOMIDAE ‣ five troglobic specires are known in Brazil, all undescribed, 4 from Pará [PA/2] and one from São Paulo state [SP/2].






PARONELLIDAE ‣ this family is the most diversified Collembola in many tropical caves but are absent in large areas like southern Sunda Islands. 29 troglobic species known in Brazil, 15 undescribed troglobic species in Brazil found in Bahia [BA/3][BA/6][BA/6], Minas Gerais [MG/3][MG/5][MG/10], Goiás [GO/1], Mato Grosso do Sul [MS/1], and São Paulo [SP/2] states.




























NEELIPLEONA
15 troglobic described species are known in this family, all in Europe and Asia, and at least one troglobic species is known in New World, a undescribed collected from SE Bahia state, E Brazil [BA/6], possibly the unique recorded troglobic Neelipleona in Western Hemisphere.
UNPLACED AT FAMILY LEVEL


10K DIPLURA ‣ following Sendra et al. (Zoological Journal of Linnean Society, 2021) and complementar sources, 170 described species are troglobic in Europe and adjacent islands except Russia and adjacent countries (110), Russia and adjacent countries (8), Africa (3, Algeria, Moroco, South Africa)[7], Lebanon (1), India (1), SE Asia (6, Myanmar, Indonesia, Papua New Guinea, Vietnam, Indonesia/Sulawesi)[22], China (5), Japan (1), Australia (7), USA/Canada (2:6/12, in Condeicampa, Holjapyx, Mixojapyx, Occasjapyx, Quapawjapyx one each, and 7 Litocampa, in Campodeidae and Japygidae, EJT)[33], Mexico (4/4, all Campodeidae), Guatemala (2, Juxtlacampa, EJT, 2026), and 10 in Brazil — one described species (Oncinocampa trajanoae Condé, 1997, from the Alto do Ribeira karst, São Paulo state)[6] and nine undescribed troglobic, in Rio Grande do Norte [RN/1], Minas Gerais [MG/1][MG/2] and Bahia [BA/2][BA/3][BA/5] states.
UNDETERMINATED FAMILY


CAMPODEIDAE


JAPYGIDAE



PROJAPYGIDAE






10L INSECTA ‣ members of 13 orders of insects occur as cave-dwellers, 12 with troglobic[4]. 313 troglobic insects occur in North America: Coleoptera (284), Dermaptera (1), Diptera (3), Hemiptera (10), Orthoptera (6), and Zygentoma (8). 202 in China[2]. Troglobic Insecta in Mexico are 65 spp. in 4 orders: Zygentoma (13), Orthoptera (4), Hemiptera (3, all Homoptera) and Coleoptera (43). Brazil has 137 spp. in eight orders: Zygentoma (8), Orthoptera (6), Hemiptera (22), Coleoptera (88), Blattodea (8), Diptera (1), Dermaptera (1) and Hymenoptera (2).
1 ZYGENTOMA
Former Thysanura. Troglobic Zygentoma widely woorldwide, 1 in Balkans[29], 4 in SE Asia[22], 1 in Australia, 8 in USA (6 Texoreddellia and two Speleonycta, both Nicoletiidae)[33], 13 in Mexico[32], 1 in Guatemala[31], 3 in Cuba[13], 1 in Argentina (ICS/vol VII, 2025), and 8 in Brazil, one described and 7 undescribed in Rio Grande do Norte [RN], Bahia [BA/2][BA/3][BA/4], Minas Gerais [MG/1], and Espírito Santo [ES/1] states, including two in Lepismatidae. When published, it will be the first oficial troglobic species in this family.
UNDETERMINED FAMILY


LEPISMATIDAE


NICOLETIIDAE






2 EPEHEMEROPTERA
A troglobic species within this order has been reported from Colombia[19], and it is provisionally accepted here. However, its validity still requires thorough confirmation in a peer-reviewed publication. If confirmed, it would represent the first described species of its group worldwide.
3 NOTOPTERA
Order formed by Manthophasmathodea and Grylloblatoidea, the former wihtout mentions about cave-dwellers. In latter, mainly species are epigean, but some species of Galloisiana and Namkungia in South Korean inhabit montane sites and many low elevation caves, wuth several reduced or eyeless species appear to be limited to cave environments. Some Grylloblatta populations are restricted to caves, morphological adaptations to the cave environment (troglomorphy) are not known, with the exception that cave-dwelling species lack pigmentation (B.Wipfler, et al, Journal of Insect Biodiversity, 2014). Besistes these reports, none troglobic is fully mentioned in literature.
4 ORTHOPTERA
76 troglobic Orthoptera are documented: Spelaeiacris tabulae Péringuey, 1916 in South Africa[7], 40 in China[2], Eutachycines cassani (Chopard, 1954) from Laos[22], 10 in Australia, six in Hawaii[33] (all Gryllidae), 6 in Mexico[5] (all Phalangospsidae), 4 in Cuba[13], 2 in Venezuela[17] and six in Brazil — all in Phalangospsidae, including one undescribed in Bahia [BA/4] state.
All cave species of Orthoptera are members of Ensifera, which are omnivorous or scavengers, and none of Caelifera, which are phytophagous. Most belong to the large families Gryllidae, Phalangopsidae, Trigonidiidae, and Rhaphidophoridae. Rhaphidiophoridae from Venezuela are the only aquatic or semi-aquatic troglobic Orthoptera (Galán & Herrera, BSVE, 2006).
PHALANGOSPSIDAE





5 BLATTARIA
38 troglobic Blattodea documented in this work, mainly saprophagous and from Old World: 5 in Africa[7], 11 in SE Asia[22], 9 in Australia, 1 in Cuba[13], 1 in Jamaica[24], 1 in Venezuela[17] (Paranocticola venezuelana Bonfils, 1987), 1 in Colombia[19], 1 in Ecuador (Ischnoptera peckorum Roth, 1988 from Galapagos Islands, Ectobiidae, Galapagos Species Database), and 8 in Brazil: Litoblatta camargoi Gutiérrez, 2005 (Blattellidae), from Bahia state (Guterrez, Solenodon, 2005), and additional 7 undescribed troglobic species, in Bahia [BA/1][BA/2][BA/3][BA/6][BA/7] and Minas Gerais [MG/2][MG/6] states.








6 PSOCOPTERA
Besides data in [4] for several possible troglobic in New World, the only clearly troglobic described representative of this order in New World is the prionoglaridid Speleopsocus chimanta Lienhard, 2010 known from a humid cave in SE Venezuela (C.Lienhard & R.L.Ferreira, Revue suisse de Zoologie, 2015). Undescribed species are reported from Colombia (1)[19] and Australia (2). Unknown troglobic Psocoptera in Brazil.
7 DERMAPTERA
There are only four troglobic in this order: Diplatys milloti Chopard, 1940 in Guinea[7], Anataelia troglobia MartÌn & OromÌ, 1988 in Canary Islands in Spain, Anisolabis howarthi Brindle, 1980 in Hawaii[33], Mesodiplatys falcifer Kamimura & Ferreira, 2018 (Diplatyidae) from Carinhanha municipality in Bahia state in dry NE Brazil, and one undescribed from Colombia[19].


8 HEMIPTERA
53 documented troglobic species are known worldwide: 4 in Spain (Subterranean Biology, 2021 | Subterranean Biology), Typhlobrixia namorokensis Synave, 1953 in Madagascar[7], 1 in China[2], 6 in SE Asia[22], 2 in Australia, (3:4/)10 in Hawaii[33] (Cixiidae-2/8, Mesoveliidae-1, Reduviidae-1), 3 in Mexico[32] (all Fulguromorpha), 2 in Jamaica[24], 1 in Colombia[19] (Cydnidae), 22 in Brazil and 1 in Argentina (Zootaxa, 2024).
Brazilian troglobic includes six families in all three suborders with cave members. The five undescribed troglobic in Brazil was 1 in Pará [PA/2], 8 in Rio Grande do Norte [RN], 2 in Bahia [BA/4][BA/5], and 1 in Mato Grosso do Sul [MS/1].
AUCHENORRHYNCHA/CIXIIDAE



AUCHENORRHYNCHA/DELPHACIDAE


AUCHENORRHYNCHA/KINNARIIDAE (VIDEO)


HETEROPTERA/DIPSOCORIDAE


HETEROPTERA/HYDROMETRIDAE



STENORRHYNCHA/ORTHEZIIDAE


9 COLEOPTERA
Approximatelly 2,500 spp. are troglobic worldwide are cited in [4]. Three families, Carabidae, Staphylinidae (including Pselaphinae and Scydmaeninae), and Leiodidae count for 98% of the diversity of troglobic and troglophilic beetles, with 1,180, 110, and 599 spp., respectively[4].
There is a considerable amount of scattered literature on Coleoptera. [4] discusses higher-level groupings, while Hlavac et al (Subterranean Biology, 2006) provides a species list for Staphylinidae. In most parts of the world, Coleoptera diversity is relatively well documented — the main exceptions are Europe, Middle East, Indian Subcontinent, NE Asia, Australia, Pacific, America Central, Caribbean, and South America from Ecuador to Argentina.
Our current numbers remain quite limited, with only 1,402 species recorded — 384 of them from Western Europe in Balkans [31], 25 in Spain and 1 in Italy (Staphylinidae non Pselaphinae, Subterranean Biology, 2006), 54 in Ariege in France (Ecography, 2006), 143 in Romania (Travaux de l’Institut de Spéologie, 2022), 22 in Africa[7], 36 in Russia and adjacent countries[21], 159 in China[2], 82 in Japan (Ecography,2006 | Subterranean Biology, 2006), 1 in Korea (Staphylinidae non Pselaphinae, Subterranean Biology, 2006), 22 in SE Asia[22], 29 in Australia, 3 in New Zealand (A Guide to the Groundwater Invertebrates of New Zealand, 2003), (9:31/)284 in USA (Anobiidae-1/1, California; Carabidae-13/193; Curculionidae-2/2, Texas; Dryopidae-2/3, Oregon and Texas; Dytiscidae-3/3, Texas; Elmidae-1/4, Texas; Leiodidae-2/22, mainly Alabama and Tennessee; Staphylinidae-7/54, mainly in Alabama; and Tenebrionidae-2/2, California and Texas)[33], 44 in Mexico (Carabidae-30, Dytiscidae-1, Histeridae-4, Leiodidae-8, Ptinidae-1)[5], 6 in Cuba[13], 4 in Jamaica[24], 1 in Guatemla[23], 3 in Venezuela (Carabidae-1, Dysticidae-1, Catopidae-1)[17], 5 in Colombia (Staphylinidae-2, Carabidae-1, Nitidulidae-1, Passalidae-1)[19], 1 in Ecuador (Ecography, 2006), 3 in Bolivia[15], 88 in Brazil, and 1 in Argentina.
(6:)89 troglobic in Brazil, being 64 undescribed: 38 in Pará [PA/2], 1 in Tocantins [TO/1], 2 in Rio Grande do Norte [RN/1], 10 in Bahia [BA/1][BA/2][BA/4][BA/5][BA/6][BA/7], 1 in Goiás [GO/1], 10 in Minas Gerais [MG/1][MG/2][MG/6][MG/10], 2 in Mato Grosso do Sul [MS/1][BR/1], and 1 in São Paulo [SP/2]. Truly troglobic species at Scarabeoidea are yet to be found in Brazil (C.M.A.Correa et al., Revista Brasileira de Entomologia, 2022).
CARABIDAE/SCARITINAE/ADISTOMINA


CARABIDAE/SCARITINAE/CLIVININA






CARABIDAE/SCARITINAE/SCHIZOGENIINA


CARABIDAE/DRYPTINAE/ZUPHIINI





















CARABIDAE/PAUSSINAE/OZAENINI


CARABIDAE/LEBIINAE/PERIGONINI


CARABIDAE/TRECHINAE


UNDETERMINED CARABIDAE










DITYSCIDAE
















EUCNEMIDAE


HISTERIDAE


HYDROSCAPHIDAE

STAPHYLINIDAE/PSELAPHINAE
















STAPHYLINIDAE/SCYDMAENINAE (including Veraphis)






UNDETERMINATED STAPHYLINIDAE








10 TRICHOPTERA
Two troglobic species within this order has been reported from Colombia[19], and it is provisionally accepted here. However, its validity still requires thorough confirmation in a peer-reviewed publication. If confirmed, it would represent the first described species of its group worldwide.
11 DIPTERA
Data about troglobic diptera is confusing because all troglobic members of this order are not or weakly modified for cave life. Several non-hematophagous Diptera are troglobic worldwide, being 2 known in Balkans[29], Sergentomyia mirabilis (Parrot & Wanson, 1939) in Tanzania[7], 2 in China [2], two Phlebotomus in Philippines[22], 4 in Australia, (3:3/)3 in USA (Spelobia tenebrarum Aldrich, 1897 in E North America, and two in Hawaii)[33], 2 in Colombia (Drosophilidae and Mycetoohilidae one each)[19], and one in N Brazil, Deanemyia maruaga Alves, Freitas & Barrett, 2008 from N Brazil, known only from Maruaga cave, in Presidente Figueiredo, Amazonas state — a parthenogenetic, hematophagous, nonvector, and can complete its full life cycle in cave[4] (Veracilda Ribeiro Alves et al., Mem. Inst. Oswaldo Cruz, 2011).


12 LEPIDOPTERA
6 described troglobic species are known in this order. Tinea microphthalma Robinson, 1980 (reduced-eyes Tineidae, Philippines) and Schrankia howarthi D. Davis & Medeiros, 2009 (Erebidae, Hawaii, SEE) presents several regressive traits: flightlessness, reduced pigmentation, and reduced eye size[4][33]. Wegneria villiersi (Viette, 1955) occur in Guinea, Africa[7], and one sp. occur in Australia. A fourth troglobic species was collected in Colombia (Angarita-Sierra, CCE/2018). Troglobic species in this order are completely unknown in Brazil.
13 HYMENOPTERA
4 ants are described as troglobic worldwide: Phaenopria cavernicola Kieffer 1906 (Tanzania)[7], Leptogenys khammouanensis Roncin & Deharveng, 2003, collected in oligotrophic habitats of a Laotian cave[4]; Aphaenogaster gamagumayaa Naka & Maruyama, 2018, from Okinawa, Japan (Naka & Maruyama, Zootaxa, 2018); and Yavnella laventa Griebenow, Moradmand & Isaia, known from Fars province, SW Iran (Z.H.Griebenow et al., Invertebrate Systematics, 2022). Three undescribed troglobic ants are known in Brazil, in Pará [PA/2], Bahia [BA/1] and Tocantins [TO/1] states.




11A PHYLUM CRANIATA/ACTINOPTERI ‣ Subterranean Fishes of the World (in December 08, 2024) lists 314 spp. of troglobic Actinopteri among 11 orders. In this source Brazil appears with 44 spp., in second diversity worldwide, after China (107, 105 in Cypriniformes, mainly in Yunnan region), and ahead Mexico (20) and USA (22). The four countries together have 193 spp. (USA and Mexico shared one spp.), more than half of the world amount of species.
Troglobitic fishes have evolved in six major clades:
OTOCEPHALA
This clades includes 4 orders, Cypriniformes (169 troglobic, all from Eurasia and Africa), Gymnotiformes (a single troglobic, endemic to E Brazil), Characiformes (two troglobic, Mexico and Brazil one each), and Siluriformes, in USA (Prietella phreatophila-1, Satan eurystomus-1, Trogloglanis pattersoni-1, the former also in Mexico), Mexico (13, Rhamdia-11, Priettella-2, one up to USA), Trinidad & Tobago (Rhamdia-1), Belize (Rhamdia-1), Venezuela (6, Ancistrus-1 Trichomycterus-4, Rhamdia-1), Colombia (10, all Trichomycterus), Ecuador (2, Astroblepus-1, Chaetostoma-1), Peru (Astroblepus-1), Bolivia (Trichomycterus-1, Phreatobius-1, also in Brazil), 37 in Brazil (one also in WC Brazil), and Argentina (Silvinichthys-1) — also Old World, in Namibia (Clarias-1), Somalia (Uegitglanis-1), India (Horaglanis-4, Kryptoglanis-2), China (2, Proliobagrus-1, Pterocryptis-1), Vietnam (Pterocryptis-1), and Thailand (Pterocryptis-1). All Brazilian troglobic fishes belongs this clade.
PERCOPSIFORMES
All 16 troglobic species of this order are endemic to USA, in Amblyopsis (2), Speoplatyrhinus (1), and Typhlichthys (13).
GOBIIFORMES
This order includes 19 troglobic species worldwide, 18 in Madagascar, Japan, Australia, Philippines, Indonesia, Papua New Guinea, and Guam, and Caecieleotris morrisi Walsh and Chakrabarty 2016, known from Mexico.
OPHIDIIFORMES
This order includes 15 troglobic worldwide in two families, Bythitidae and Dinematichthyidae, 11 of these in Cuba and Bahamas, two in Mexico, one in Galapagos and one in Indonesia.
SYNBRANCHIFORMES / ANABANTOIDEI
Two orders in this clade includes troglobic, Anabanthiformes (a single species, known from Kerala state, S India) and Synbranchiformes, with 10 spp., all in Synbranchidae (six in India, Mexico, Venezuela, Cameroon and Australia).
PERCIFORMES
This order includes only 3 troglobic species, all in Cottus, from Pensylvannia and Missouri, E USA.

PHYLOGENY OF ACTINOPTERI HIGHLIGHTING THE ORDERS WITH TROGLOBITIC SPECIES IN BRAZIL (GREEN), MEXICO (YELLOW), THE USA (BROWN), AND THE REST OF THE WORLD (GRAY)
Of the 45 troglobic fish species cited for Brazil in SFW (44 nominally + Phreatobius undescribed species Aripuana), six are excluded, and the remaining 39 are added to one undescribed in Bahia [BA/4], totaling 40 species accepted in this article, in Characiformes (1:1/1), Gymnotiformes (1:1/1) and Siluriformes (4:10/38). The six species rejected here are Isbrueckerichthys alipionis Derijst 1996 (Loricariidae, isolated cave population of an otherwise epigean species, SEE), and five undescribed Phreatobius (Heptapteridae, being four in Rio Negro basin and one in Aripuanã river basin — full details in Cunha, Thesis, 2018).
Although Poecilia mexicana Steindachner, 1863 (Poeciliidae, Cyprinodontiformes) and Astyanax aeneus Günther,
1860 (Characidae, Cypriniformes) are listed in the SFW, we reject its troglobic status here, as it is a species with epigean populations (Wikipedia) for both and is therefore excluded from the scope of this article. The figures below already reflect this adjustment.

COLOR GRADIENT OF OTOCEPHALA TROGLOBIC DIVERSITY IN NW, AND DATA FOR THE OTHER FIVE TROGLOBIC LINEAGES IN THE NW
All Brazilian troglobic fishes are quite small, with only three species exceeding 100 mm. A manual search for size data revealed: Pimelodella kronei (20.2 cm, PlanetCatfish), Rhamdia enfurnada (147 mm, SEE), and Eigenmannia vicentespelaea (up to 123 mm, SEE). P. kronei holds two remarkable distinctions: it is the oldest described troglobic species in Brazil (1907) and, based on the sizes listed above — and assuming no Brazilian troglobic invertebrate exceeds 100 mm — it is also the largest troglobic in the country.
Neolissochilus pnar Dahanukar, Sundar, Rangad, Proudlove & Raghavan, 2023 (Cyprinidae) endemic to NE India is the largest troglobtic fish worldwide, possibly the largest of all troglobic (Novotaxa).
All genera with troglobic fish in South America have them in Brazil except (only troglobic listed) Astroblepus (2, Astroblepidae, Ecuador and Peru one troglobic each), Chaetostoma (1, Loricariidae, Ecuador), Silvinichthys (1, Trichomycteridae, Argentina), Ogilbia (1, Dinematichthyidae, Galapagos) and Synbranchus (1, Synbranchidae, Venezuela).
In New World, exclusively underground endemic genera are Trogloglanis and Satan, both monotypic of Ictaluridae endemic to the USA, Amblyopsis, Triglichthys and Speoplatyrhynus, all of Amblyopsidae, which is, as a whole, an endemic family of the USA; and Stygichthys (Characidae), endemic to SE Brazil. No other country has entirely underground endemic genera of fish. Only Prietella phreatophila Carranza, 1954 (N Mexico/S USA) and Phreatobius sanguijuela (WC Brazil/NE Bolivia) are noN-national endemisms.
CHARACIFORMES
Troglobic in this order includes only two Characidae: Astyanax jordani Hubbs & Innes, 1936, from San Luiz Potosi and Tamaulipas in Mexico, and Stygichthys typhlops Brittan & Böhlke, 1965, known from two wells from Jaiba and Verdelêndia municipalities, N Minas Gerais state, E Brazil.


GYMNOTIFORMES
Eigenmannia vicentespelaea Triques, 1996 (Sternopygidae) is the unique stygobitic fish in this order worldwide, known only from São Vicente I cave in São Domingos, NE Goias state, Brazil.


SILURIFORMES
Speciose order with 82 troglobic species worldwide in 11 families, four of these has troglobic members in Brazil.
CALLYCHTHYIDAE


HEPTAPTERIDAE


















LORICARIIDAE

TRYCHOMYCTERIDAE


















11B PHYLUM CRANIATA/AMPHIBIA ‣ 13 amphibians worldwide are troglobic, in three genera[2][33]: Eurycea (Plethodontidae), with 9 spp., from Texas, Arkansas, Kansas, Missouri, Oklahoma, Florida and Georgia; Gyrinophilus (Plethodontidae), with 3 spp. from Alabama, Georgia, Tennessee and West Virginia; and Proteus (Proteidae), with P. anguinus Laurenti, 1768, known from Dinaric Mountains in NE Italy, Slovenia, Croatia, and Bosnia-Herzegovina, the only stygobitic salamander in Europe and unique member of their family in continent, reaches a length of more than 25 cm, making the second largest stygobitic (and troglobic) known anywhere after Neolissochilus pnar (Actinopteri) from India. No cave salamander in Mexico[5].
[2] cites one cave-dwelling Anura in China, however is fully rejected here as troglobic, being excludes in this count. V.F.Sperandei et al. (Biota Neotropica, 2024) discuss Anura in caves in Brazil, citing 54 species that have already been collected in caves. Oreobates antrum Vaz-Silva, Maciel, Andrade and Amaro, 2018 has only been found inside caves, in the states of Goiás and Tocantins (A.Motta et al, Herpetology Notes, 2020). However, there are no known records of troglobic Anura worldwide. Some reports among Bokermannohyla in Minas Gerais state are found in ICMBIO/News.
LAST UPDATES
May 14, 2026 ‣ adjustments in Platyhelminthes, involving the exclusion of two species previously treated as undescribed, but now tentatively considered to correspond to two species described subsequently and already included in the list.
April 1, 2026 ‣ added new troglobic species: Ceriochernes spelaeofoliaceus, C. foliaceospeluncarum (Carinhanha/BA and Prudente de Morais/MG, 2025), Pseudochthonius cesarioensis, P. gonssaloensis (Campo Formoso, BA, 2025), Eukoenenia pili, E. renatae, E. sp.nov.1 (Mariana/Santa Barbara, MG, 2026), Troglorhopalurus araras (Ituaçu, BA, 2026), Pandirodesmus zogbieae (PA, Aveiro, 2025), Newportia eleonorae (Bodoquena, MS, 2025), Spelaeogammarus quilombola (BA, Morro do Chapéu, 2025), Hyalella karajas, H. amazonica, H. nebula, H. uva (PA/PA/MG/RS, Canaã dos Carajás, Brumadinho, Santa Tereza, 2025), Aegla clotho, A. atropos (SP, Iporanga, Guapiara, 2024), Brasilana spelaea (Quixere, Felipe Guerra, Baraúna, Governador Dix-Sept Rosado, RN/CE, 2025), Circoniscus boitata (Itacarambi, MG, 2025), Kaatecarcinia apina, K. amazonica, K. karaja (Parauapebas, Curionópolis, PA, 2026), Novamundoniscus anophthalmus, Trichorhina dimorphica, Pectenoniscus bilobulatus (MS/BA/BA, Bodoquena and Coribe, 2025), Iansaoniscus aykuara (Rio Pardo de Minas, MG, 2025), Spelaeodelphax nexus (Carinhanha, BA, 2026), Pintalia magnaepiprocti (Presidente Olegário, MG, 2025), and expansion of distribution of Xangoniscus lapaensis and Pectenoniscus pankaru up to Carinhanha (López-Orozco, CM et al., Annales Zoologici, 2025).
April 1, 2026 ‣ incorporation of data on the distribution of Pseudochthonius biseriatus (Bedoya-Roqueme et al, Zootaxa, 2023, now in Pernambuco state), exclusion of Gabuinillo aridicola (Isopoda/Armadillidae, Souza et al., Zootaxa, 2010) and Ancistrus cryptophthalmus (due to epigean populations, Alves, BVA et al, International Journal of Speleology, 2013), and updating of records of troglobic from Rio Grande do Norte (Bento & Cruz, CaverRNas: o Carste Potiguar, 2025), in addition to a complete and thorough revision, correction, and update of the page’s text and code, as well as of the physical files that enable the maintenance and updating of this content.
October 20, 2025 ‣ addition of 122 undescribed troglobic species (including first record of a troglobic Schizomida in Brazil) collected in various inventories in the Carajás Range, Pará state, compiled by Trevelin et al. (Ecological Indicators, 2019).
October 13, 2025 ‣ addition of nine undescribed species for Pará, originating from Paraíso Cave in Aveiro (SEE).
October 12, 2025 ‣ numerical updates and a complete revision were carried out for the USA (including Hawaii and Alaska) and Canada (Niemiller, M.L. et al., Biodiversity and Conservation, 2025), along with partial updates for South Korea (SEE | SEE), Argentina (1 Diplopoda, SEE), and Mexico (1 Opiliona, SEE).
June 14, 2025 ‣ corrections in Actinopteri and inclusion of Endecous spelaeus (Orthoptera, BA, SEE).
May 29, 2025 ‣ addition of Pseudochthonius maquinensis, P. urubuquaqua (ZOOTAXA, 2025), Spelaeogammarus rafaelae, S. lundi (Taxonomy, 2025), Circoniscus paradisus (Nauplius, 2025), and Brasileirinho sergipanus (Zootaxa, 2025).
March 22, 2025 ‣ addition of 20 undescribed species of Padre Cave, Santana municipality, Bahia (SEE), and 13 undescribed species for Paraná state (SEE).
March 16, 2025 ‣ addition of one Hydroscaphidae, collected in Jardim municipality, Mato Grosso do Sul (SEE).
January 05, 2025 ‣ addition of Girardia patiensis and G. alba (BA, Platyhelminthes, Zoologischer Anzeiger, 2025).
December 26, 2024 ‣ addition of 7 spp. of Xangoniscus (Isopoda) from Bahia: X. paiabare, X. tymaopeba, X. ykanhema, X. puku (SEE), Xangoniscus antiquus, X. chaimowiczi, X. jonasi (SEE), subsequent updates, and upadates and correction among Bahian municipalities; addition of two Gastropoda from Carinhanha, Bahia (SEE), Cryptops didi from Iporanda, São Paulo state (SEE); Pseudochthonius limettioides for Caeté, Mariana and Santa Barbara in Minas Gerais (SEE), P. aware for Santa Maria Vitória in Bahia (SEE), and Paleotoca diminas for Presidente Prudente in Minas Gerais (SEE), and addition of 13 undescribed species after (G.A.S. Vaz et al, Acta Oecologica, 2024), knwon from Pedro Cassiano cave, Carinhanha, Bahia state.
December 15, 2024 ‣ update and revision, text and terminology standardization, HTML code correction, topic structuring, language smoothing, species authorship, visual optimization, and information accuracy throughout the post.
November 13, 2024 ‣ corrections and simplification of the writing, code optimization, reordering of data about Indonesia, Australia, inclusion of data from Africa and Croatia.
April 23, 2024 ‣ exclusion of Endecous apterus and E. peruassuensis (Orthoptera), rejected in Carvalho, P.H.M. et al. (Zootaxa, 2023).
25∙04∙2024 ‣ addition of 11 new described species: Pinelema elinae (BA), Loxosceles boqueirao (BA), L. bodoquena (MS), Spaeleoleptes gimli (BA), Cayenniola albaserrata (BA), Trichorhina baiana (BA), Ctenorillo iuiuensis (BA), Circoniscus mendesi (PA), Circoniscus xikrin (PA), Kadiweuoniscus rebellis (MS) and Endecous vitreus.
22∙01∙2024 ‣ inclusion of 10 new undescribed species in Aguas Claras System, Carinhanha (BR), in Araneae, Coleoptera, Diplura, Hemiptera and Dilplopoda (SEE).
22∙01∙2024 ‣ exclusion of Pseudonannolene canastra (Diplopoda), synonimized under P. ambuatinga, with ratification of accounting on several topics and readaptation of the P. ambuatinga map (SEE).
20∙10∙2023 ‣ inclusion of Caraiboscia jabutiensis (Isopoa, MT, SEE), Gabunillo enfurnado, Venezillo moreirai, V. limai (Isopoda, BA, SEE) and Spelaeometra hypogea (Hemiptera, BA, SEE).
16∙10∙2023 ‣ of records of undescribed troglobic species in three regions: Lapa Nova, Vazante, MG (6 spp., SEE), Lagoa Santa, MG (4 spp., SEE) and Presidente Olegário, MG (3, SEE).
13∙10∙2023 ‣ inclusion of 5 new Pectenoniscus (Isopoda, SEE) and Pseudochthonius lubueno (SEE).
10∙09∙2023 ‣ updates in undescribed species in Bahia state, with a inclusion f 30 news undescribed (Gallão et al., Diversity, 2023); inclusion of Plutogeophilus jurupariquibaba, former undescribed (Chilopoda, [SP/2], SEE); inclusion of Benthana alba, Benthanoides amazonicus, B. tarzan (Cardoso & Ferreira, Zootaxa, 2023), Ferricixius michaeli and F. goliathi (Ferreira et al., Zootaxa, 2023).
25∙07∙2023 ‣ updates in troglobic Hirundina and Polychaeta at Annelida.
20∙07∙2023 ‣ updates in Amblypygi, with a remarkable new record in Colombia.
30∙06∙2023 ‣ updates about troglobic sponges, and troglobits in Mexico, and inclusion of Phretobius sanguijuela, with general updates in Heptapteridae.
19∙06∙2023 ‣ inclusion of Endecous infernalis, former undescribed [BA/1](SEE).
09∙06∙2023 ‣ add three new species: Pseudochthonius koinopoliteia, former undescribed [BA/1], P. diamachi, P. pali (Prado & Ferreira, Zootaxa, 2023).
03∙01∙2023 ‣ updates in Collembola, inclusion of Iandumoema cuca, I. gollum, I. stygi (Opiliones), Dobrodesmus mirabilis (Dobrodesmidae), Ardistomis ferreirai (Carabidae), Metopioxys carajas, Oxarthrius aurora, O. inexpectatus (Staphylinidae),Leptus sidorchukea (Erythraeidae), Tonton itabirito, T. matodentro (Microstigmatidae), Pararrhopalites sideroicus (Sminthuridae), Onciurosoma troglobium (Paradoxosomatidae), Loxocelis troglobia (Sicariidae), Brasilomma enigmatica, Indiani gaspar, Paracymbiomma bocaina, P. pseudocaesus (Prodidomidae), Ochyrocera dorinha, O. rosinha, O. ungoliant, Speocera babau, S. pinima, S. piquira (Ochyroceratidae), Charinus cearensis, C. diamantinus, C. euclidesi, C. puri, C. renneri (Charinidae), Aegla charon (Aeglidae), Lavajatus moroi (Subulinidae), and exclusion of Troglorhopalurus lacrau (Scorpiona), Isoctenus corymbus (Ctenidae), Lygromma ybyguara (Prodidomidae) and Spiripockia umbraticola (Pomatiopsidae), by indications in [6], and inclusion of Eukoenenia mocororo.
08∙08∙2022 ‣ conclusion of a huge revision of all data from this page, inc. several notes for Mexico and USA.
03∙07∙2022 ‣ a huge revision, including c. 280 new species in list, mainly undescribed.
12∙06∙2022 ‣ inclusion of Perigona spelunca (Carabidae).
26∙11∙2021 ‣ inclusion in the list of troglobic in Brazil of 6 spp. de Matta (Araneae) and updated the text to include this addition.